Pregnancy 101
Prenatal Screening Tests
The field of prenatal screening has rapidly evolved over the last few years, and will likely continue to change in the future. There are many different types of screening tests available during pregnancy. It is important to know that screening tests will only tell you about the chance of a medical condition. Screening tests will not give you a ‘yes’ or ‘no’ answer. It is also important to know that prenatal screening tests are always optional. You never have to undergo any prenatal testing if you do not wish to. Anyone who is having prenatal testing should have access to all of the necessary information to make an informed decision about what is right for them and their family.
Prenatal ultrasound, also known as sonogram, is an imaging technique that is used to look at a fetus during the pregnancy. Ultrasound involves using sound waves that reflect off of the different parts of the baby to generate a picture. This picture can be used to take measurements, which can be used to verify how far along a pregnancy is and to monitor growth throughout the pregnancy. Ultrasound can also be used to screen for birth defects, such as spina bifida, heart defects, and cleft lip.
It is fairly common to have at least one ultrasound during pregnancy. However, some women may have an earlier ultrasound or more ultrasounds if they have bleeding, pain, or other concerns are identified on an initial ultrasound.
Currently, there is no reliable evidence that ultrasound is harmful to a developing baby. For example, there have been no associations between ultrasound and birth defects, childhood cancer, or developmental delays later on in life.
However, according to the American College of Obstetricians and Gynecologists patient information on ultrasound, it is possible that harmful effects could be identified down the road. Their recommendation is that ultrasounds be performed only for medical reasons by qualified health care providers.
There are generally two types of ultrasounds that are used during pregnancy to screen for genetic conditions and birth defects: the first trimester ultrasound, and the anatomy ultrasound.
First Trimester Ultrasound
The first trimester ultrasound is usually done in the first trimester (from conception up until about 14 weeks), and the baby is small at this time, only a few inches in length. Although the fetus is still very little, there are some important things that this ultrasound can tell us:
- It can verify the presence of a viable pregnancy
- It can estimate how far along the pregnancy is
- It can see if there is more than one fetus (twins, triplets, etc)
- It can measure a pocket of fluid behind the fetus’ neck called the nuchal translucency (NT)
- If the NT measurement is larger than expected, it can increase the risk for chromosome conditions, such as Down syndrome, as well as heart defects
A normal first trimester ultrasound does not guarantee a healthy baby, but it does lower the chance for certain birth defects and genetic conditions.
Anatomy/Level 2 Ultrasound
The anatomy (or level 2) ultrasound, usually done around 20 weeks, is where a lot of measurements are taken. The fetus is checked out from head to toe, and measurements are taken of its head, limbs, and abdomen. During this screening test, the doctor will also look at important organs, such as the heart, the brain, and the kidneys. This is usually the ultrasound where the gender of the baby can be determined. The anatomy ultrasound can also make sure the placenta looks healthy, and that there is the right amount of amniotic fluid around the baby. Much like the first trimester ultrasound, a normal anatomy ultrasound does not guarantee a healthy baby, but it can lower the risk for certain birth defects and genetic conditions.
While most anatomy ultrasounds are completely normal, there are occasions when the doctor may find something that looks different on an ultrasound. These difference can be broken down into two categories: structural abnormalities (birth defects) and minor markers.
First trimester screening (FTS) is a test that can give more information on the chances that a pregnancy has Down syndrome (trisomy 21) and trisomy 18 (some labs also offer screening for trisomy 13). FTS is generally performed between 11 and 14 weeks of the pregnancy, and is made up of two parts: a blood draw from mom and an ultrasound. The blood draw measures two proteins that are in mom’s blood that are coming from the pregnancy: pregnancy-associated plasma protein-A (PAPP-A), and human chorionic gonadotropin (hCG).
- Pregnancy-associated plasma protein-A (PAPP-A): PAPP-A is produced by the placenta and crosses over into mom’s blood stream. Lower levels of PAPP-A increase the chances of Down syndrome or trisomy 18, and can also be associated with poor pregnancy outcomes (miscarriage, low birth weight, and high blood pressure/preeclampsia). However, keep in mind that most pregnancies that have low PAPP-A levels go on to produce healthy babies.
- Human chorionic gonadotropin (hCG): hCG is also produced by the placenta and crosses over into mom’s blood stream. hCG tends to be higher in pregnancies with Down syndrome and lower in pregnancies with trisomy 18.
The first trimester ultrasound measures the fetus to verify dating, as well as pocket of fluid behind the baby’s neck called the nuchal translucency (NT). There should be some fluid in this area, but if the fluid measures larger than expected, it increases the chance for Down syndrome, trisomy 18, and other birth defects.
The lab uses the measurements from the ultrasound and the blood draw, combined with information about the mom (age, weight, ethnicity, etc) to calculate a risk number. The results will be broken down by each condition that was screened for. For example, a first trimester screen result may say that the chance for Down syndrome is 1 in 5,000, and the chance for trisomy 18 is 1 in 10,000. This means that if there were 5,000 women that had the exact same test result, only 1 would have a child with Down syndrome. Likewise, if there were 10,000 women with the exact same test result, only 1 would have a child with trisomy 18.
A low-risk, or negative, FTS means the chances are reduced, but this test does not rule out any medical conditions. Similarly, a FTS result that shows an increased chance does not mean that the pregnancy has that condition. If your results show an elevated risk, you may be offered further testing, such as a diagnostic prenatal test. It’s important to remember that most people who have a ‘positive’ or ‘high-risk’ FTS will go on to have healthy babies.
People choose either to do or not to do prenatal genetic screening for many reasons. It is important to remember that this testing is voluntary and is not required.
Your results are high-risk. Now what?
If you had a FTS done with your primary OB-GYN, they may refer you to a high-risk pregnancy doctor called a perinatologist. You may also be referred to meet with a genetic counselor. These visits are designed to gather more information, but also to make sure you have a clear understanding of everything that is going on. It is also important during these visits to talk about the risks, benefits, and limitations of different options for further testing, including cell-free DNA, chorionic villus sampling (CVS), or amniocentesis. The choice of whether or not to undergo further testing is yours, and further testing is always voluntary.
Your results are low-risk. Now what?
A low-risk test result means the chance that the pregnancy has the conditions that were screened for is reduced, but not eliminated. Depending on the lab, between 80-95% of pregnancies that have Down syndrome and trisomy 18 will have a positive FTS, which means that approximately 5-20% will have low-risk FTS results. The chance that a pregnancy that does not have Down syndrome or trisomy 18 will have a high-risk test result, or a false positive, is usually between 3-5% (depending on the lab).
How do I decide whether to get FTS or not?
If you’re on the fence about whether or not to pursue FTS, it may be helpful to consider the following:
- If the FTS shows a high risk for a condition, would you pursue additional testing, such as CVS or amniocentesis?
- If not, would you be ok waiting until the baby is born to know for sure if they are affected?
- Would you want this kind of information to be better prepared?
- Would you consider doing anything differently if you knew a pregnancy was affected with one of these conditions, such as placing the baby up for adoption or not continuing the pregnancy?
- Does more information with the possibility of uncertainty make you nervous or anxious?
It is important to talk with your medical provider or a genetic counselor if you have further questions or concerns.
Second trimester screening (STS), also called quad screen or triple screen, is a test that can tell the chances that a pregnancy has Down syndrome (trisomy 21), trisomy 18, and open neural tube defects (ONTDs). STS is generally done between 15 and 22 weeks of the pregnancy, and consists of a blood draw taken from mom. The triple screen measures three hormones and proteins that are in mom’s blood that are coming from the pregnancy: human chorionic gonadotropin (hCG), unconjugated estriol (uE3), and alpha-fetoprotein (AFP).
- Unconjugated estriol (uE3): uE3 is made by both the baby’s liver and the placenta. Levels of this hormone rise throughout the pregnancy. Babies with Down syndrome or trisomy 18 tend to have lower levels of uE3.
- Human chorionic gonadotropin (hCG): hCG is hormone that is also produced by the placenta and crosses over into mom’s blood stream. hCG tends to be higher in pregnancies with Down syndrome and lower in pregnancies with trisomy 18.
- Alpha-fetoprotein (AFP): AFP is a protein produced in the baby’s liver during the second trimester of pregnancy. The levels of AFP generally increase the further into the pregnancy you are. AFP levels tend to be higher when the baby has an open neural tube defect (ONTD), such as spina bifida. With ONTDs, there is an opening in the skin and AFP has another ‘escape route’ out. AFP levels tend to be lower in pregnancies that have Down syndrome or trisomy 18.
The quad screen analyzes the three previous substances, as well as inhibin-A.
- Inhibin-A: Inhibin-A comes from the placenta. Levels of this hormone in mom’s blood remain relatively constant through weeks 15-18 of the pregnancy. Pregnancies with Down syndrome tend to have higher than average levels of inhibin-A, while pregnancies with trisomy 18 tend to have lower levels of inhibin-A.
The lab uses information from the blood draw and about the mom (age, weight, ethnicity, etc) to calculate a risk number. The results will be broken down by each condition that was screened for. For example, a second trimester screen result may say that the chance for Down syndrome is 1 in 5,000, the chance for trisomy 18 is 1 in 10,000, and the chance for ONTDs is 1 in 3,000. This means that if there were 5,000 women that had the exact same test result, only 1 would have a child with Down syndrome. Likewise, if there were 10,000 women with the exact same test result, only 1 would have a child with trisomy 18. And if there were 3,000 women with the exact same test results, only 1 would have a child with an ONTD.
A low-risk, or negative, STS means the chances are reduced, but this test does not rule out any medical conditions. Similarly, a STS result that shows an increased risk does not mean that the pregnancy has that condition. If your results show an elevated risk, you may be offered further testing, such as a diagnostic prenatal test. It’s important to remember that most people who have a ‘positive’ or ‘high-risk’ STS will go on to have healthy babies.
People choose either to do or not to do prenatal genetic screening for many reasons. It is important to remember that this testing is voluntary and is not required.
Your results are high-risk. Now what?
If you had a STS done with your primary OB-GYN, they may refer you to a high-risk pregnancy doctor called a perinatologist. You may also be referred to meet with a genetic counselor. These visits are designed to gather more information, but also to make sure you have a clear understanding of everything that is going on. It is also important during these visits to talk about the risks, benefits, and limitations of different options for further testing, including cell-free DNA or amniocentesis. The choice of whether or not to undergo further testing is yours, and further testing is always voluntary.
Your results are low-risk. Now what?
A low-risk test result means the chance that the pregnancy has the conditions that were screened for is reduced, but not eliminated. Depending on the lab, between 75-85% of pregnancies that have Down syndrome and trisomy 18 will have a positive STS, which means that approximately 15-25% will have low-risk STS results. The chance that a pregnancy that does not have Down syndrome or trisomy 18 will have a high-risk test result, or a false positive, is usually between 5-8% (depending on the lab).
How do I decide whether to get STS or not?
If you’re on the fence about whether or not to pursue STS, it may be helpful to consider the following:
- If the STS shows a high risk for a condition, would you pursue additional testing, such as amniocentesis?
- If not, would you be ok waiting until the baby is born to know for sure if they are affected?
- Would you want this kind of information to be better prepared?
- Would you consider doing anything differently if you knew a pregnancy was affected with one of these conditions, such as placing the baby up for adoption or not continuing the pregnancy?
- Does more information with the possibility of uncertainty make you nervous or anxious?
It is important to talk with your medical provider or a genetic counselor if you have further questions or concerns.
Sequential screening (SS) is a test that can tell the chance that a pregnancy has Down syndrome (trisomy 21), trisomy 18, and open neural tube defects (ONTDs). The first part of the SS is generally performed in the first trimester and consists of a blood draw from mom and an ultrasound. Part two consists of a second blood draw from mom.
The first part of the sequential screen is similar to the first trimester screen (FTS), and the second part is similar to second trimester screening (STS). The first part of the sequential measures two hormones and proteins that are in mom’s blood that are coming from the pregnancy: pregnancy-associated plasma protein-A (PAPP-A), and human chorionic gonadotropin (hCG).
- Human chorionic gonadotropin (hCG): hCG is also produced by the placenta and crosses over into mom’s blood stream. hCG tends to be higher in pregnancies with Down syndrome and lower in pregnancies with trisomy 18.
- Pregnancy-associated plasma protein-A (PAPP-A): PAPP-A is produced by the placenta and crosses over into mom’s blood stream. Lower levels of PAPP-A increase the chances of Down syndrome or trisomy 18, and can also be associated with poor pregnancy outcomes, (miscarriage, low birth weight, and high blood pressure/preeclampsia). However, keep in mind that most pregnancies that have low PAPP-A levels go on to produce healthy babies.
The results of the blood test is combined with the first trimester ultrasound, which measures the fetus to verify dating as well as pocket of fluid behind the baby’s neck called the nuchal translucency (NT). The lab uses a formula that includes the information from the blood work as well as the NT measurement to come up with a risk number. The second part of the sequential screen measures hCG again, as well as:
- Inhibin-A: Inhibin-A comes from the placenta. Levels of this hormone in mom’s blood remain relatively constant through weeks 15-18 of the pregnancy. Pregnancies with Down syndrome tend to have higher than average levels of inhibin-A, while pregnancies with trisomy 18 tend to have lower levels of inhibin-A.
- Unconjugated estriol (uE3): uE3 is made by both the baby’s liver and the placenta. Levels of this hormone rise throughout the pregnancy. Pregnancies with Down syndrome or trisomy 18 tend to have lower levels of uE3.
- Alpha-fetoprotein (AFP): AFP is a protein produced in the baby’s liver during the second trimester of pregnancy. The levels of AFP generally increase the further into the pregnancy you are. AFP levels tend to be higher when the baby has an open neural tube defect (ONTD), such as spina bifida. With ONTDs, there is an opening in the skin and AFP has another ‘escape route’ out. AFP levels tend to be lower in pregnancies that have Down syndrome or trisomy 18.
The lab uses information from the blood draws and about the mom (age, weight, ethnicity, etc) to calculate a risk number. The results will be broken down by each condition that was screened for.
The first part of the sequential screen will usually give the chance for Down syndrome and trisomy 18. The results do not tell you ‘yes’ or ‘no’, but tell you the chance that the pregnancy has that condition. For example, the first part of the sequential screen may say that the chance for Down syndrome is 1 in 5,000, and the chance for trisomy 18 is 1 in 10,000. This means that if there were 5,000 women that had the exact same test result, only 1 would have a child with Down syndrome. Likewise, if there were 10,000 women with the exact same test result, only 1 would have a child with trisomy 18.
The second part of the sequential screen will give you an updated risk for Down syndrome and trisomy 18, as well as the risk for ONTDs.
A low-risk, or negative, SS means the chances are reduced, but this test does not rule out any medical conditions. Similarly, a SS result that shows an increased risk does not mean that the pregnancy has that condition. If your results show an elevated risk, you may be offered further testing, such as a diagnostic prenatal test. It’s important to remember that most people who have a ‘positive’ or ‘high-risk’ SS will go on to have healthy babies.
People choose either to do or not to do prenatal genetic screening for many reasons. It is important to remember that this testing is voluntary and is not required.
If you had a SS done with your primary OB-GYN, they may refer you to a high-risk pregnancy doctor called a perinatologist. You may also be referred to meet with a genetic counselor. These visits are designed to gather more information, but also to make sure you have a clear understanding of everything that is going on. It is also important during these visits to talk about the risks, benefits, and limitations of different options for further testing, including cell-free DNA or amniocentesis. The choice of whether or not to undergo further testing is yours, and further testing is always voluntary.
A low-risk test result means the chance that the pregnancy has the conditions that were screened for is reduced, but not eliminated. Depending on the lab, between 90-96% of pregnancies that have Down syndrome and trisomy 18 will have a positive SS, which means that approximately 4-10% will have low-risk SS results. The chance that a pregnancy that does not have Down syndrome or trisomy 18 will have a high-risk test result, or a false positive, is usually between 3-5% (depending on the lab).
If you’re on the fence about whether or not to pursue SS, it may be helpful to consider the following:
- If the SS shows a high risk for a condition, would you pursue additional testing, such as CVS or amniocentesis?
- If not, would you be ok waiting until the baby is born to know for sure if they are affected?
- Would you want this kind of information to be better prepared?
- Would you consider doing anything differently if you knew a pregnancy was affected with one of these conditions, such as placing the baby up for adoption or not continuing the pregnancy?
- Does more information with the possibility of uncertainty make you nervous or anxious?
It is important to talk with your medical provider or a genetic counselor if you have further questions or concerns.
Cell-free DNA (cfDNA) is a screening test that can be performed any time after 10 weeks of pregnancy that screens for common chromosome conditions (Down syndrome, trisomy 18, and trisomy 13), as well as sex chromosomes (XX and XY), which can also incidentally tell the sex of the baby.
cfDNA has also been called non-invasive prenatal screening (NIPS), non-invasive prenatal testing (NIPT), and by several brand names, such as Harmony, Panorama, MaterniT21, Verifi, and InformaSeq. cfDNA will not tell you ‘yes’ or ‘no’ about these conditions, but can tell you whether there is an increased chance for the pregnancy to have these chromosome conditions. If the results say there is an increased risk, you may be offered diagnostic tests, such as chorionic villus sampling (CVS) or amniocentesis. These diagnostic tests can provide ‘yes’ or ‘no’ answers, but come with a risk of miscarriage. Because of this, diagnostic tests are optional and are never anything someone has to do.
The conditions that are screened for can vary in severity, from little to no medical issues to severe or life-limiting health complications. Which conditions are screened for also vary from lab to lab, but most labs screen for Down syndrome, trisomy 18, trisomy 13, fetal sex, and sex chromosome differences. Most of these conditions do not run in families, but the chance of chromosome conditions, such as Down syndrome, are higher in women who will be 35 or older when the baby is born.
If a screening test, such as cfDNA, were to show an increased risk for a condition, a diagnostic test, such as amniocentesis or chorionic villus sampling (CVS), would be available to provide more definitive information. It is also important to remember that if a screening test shows a low risk, the chance the pregnancy has those conditions is reduced but not eliminated.
It is important to discuss your cfDNA results with a doctor or genetic counselor to understand what the results mean for your pregnancy, and make sure you have all of the information necessary to decide on the best next steps for you and your family.
cfDNA Testing: How does it work?
Our DNA is inside nearly every cell of our body, and is the instruction manual for how everything in our body grows and functions. Our cells are continuously dividing to create new cells. As cells break down, the DNA inside the cell is released into the blood as fragments or pieces of DNA. These DNA fragments in the blood are known as cell-free DNA.
All pregnant women will have DNA fragments in their blood (some that are her DNA, and some that is from the placenta). The placenta develops during pregnancy and provides oxygen and nutrients to the developing baby. The placenta has the same genetic information as that of the developing baby approximately 98-99% of the time.
However, approximately 1-2% of the time, the genetic makeup of the placenta and that of the developing baby are not the same (called confined placental mosaicism). This can lead to false positive (the results are high-risk but the fetus does not actually have the condition) and false negative results (the results are low-risk but the fetus actually has the condition). This is one reason why tests such as cfDNA are considered screening tests; they cannot tell you ‘yes’ or ‘no’ for sure if your pregnancy has any of these conditions. It can only tell you if there is an increased or decreased chance.
When it comes to how the test is run, there are slight differences in the technologies that various labs may use. In general, regardless of the technology used, the lab is looking for differences in the overall amount of chromosome material in mom’s blood.
For example, if the testing finds an increased amount of material that is from chromosome 21, then the test result would say that there is an increased risk that the pregnancy has Down syndrome. Again, the result from a cfDNA test would never be able to tell you ‘yes’ or ‘no’, but can help give you more information about the risk for the conditions tested in your pregnancy.
cfDNA Testing: What conditions are screened for?
cfDNA started as a screening test for Down syndrome, but over time the list of conditions that can be screened for is expanding. Most labs now provide screening for Down syndrome, trisomy 18, and trisomy 13, as well as the sex chromosomes (X and Y). Down syndrome, trisomy 18, and trisomy 13 are all caused by an extra chromosome.
Screening for sex chromosomes can help predict the sex of the baby, but it can also screen for extra or missing sex chromosomes. These sex chromosome differences can vary widely from mild with no notable physical or developmental differences to severe and life-limiting in rare cases. There are four more common sex chromosome differences that are screened for: Monosomy X/Turner syndrome (45,X), Triple X (47,XXX), Klinefelter syndrome (47,XXY), and Jacobs syndrome (47,XYY).
Some labs have recently started offering screening for a set of genetic conditions called microdeletion syndromes. Microdeletions are when a small piece of a chromosome is missing, rather than an entire chromosome. There are currently no medical guidelines in place to support using cfDNA to screen for microdeletion syndromes, but there may be instances where this information could be helpful. Some microdeletion conditions that labs are screening for are:
- 22q11 deletion syndrome
- 1p36 deletion syndrome
- Angelman syndrome
- Prader-Willi syndrome
- Cri-du-chat
- Wolf-Hirschhorn syndrome
- Jacobsen syndrome
- Langer-Giedion syndrome
Because there is great variation in the conditions that may be screened for, it is important to discuss these options with your provider to make a decision that is best for you and your family.
cfDNA Testing vs. Traditional Screening
There are pros and cons to both cfDNA screening and traditional screening, such as first trimester screening, second trimester screening, sequential screening, and ultrasound.
For some conditions, particularly Down syndrome, cfDNA has been shown to be a more accurate screening test than traditional screening tests. cfDNA may also potentially screen for more chromosome conditions than traditional testing.
However, traditional screening can assess for conditions that would not be found with cfDNA screening, such as open neural tube defects (ONTDs). Traditional screening tests can also detect an increased risk for pregnancy complications, such as preeclampsia, preterm labor, and growth restriction.
cfDNA has the benefit of being able to be done sooner in pregnancy, often as early as 10 weeks. cfDNA is also able to test for sex as well as sex chromosome differences (more or fewer X or Y chromosomes than expected). Some labs offer screening for other genetic conditions, such as microdeletion syndromes, for which there is limited available data.
It is important to discuss with your provider what your cfDNA test will screen for, and to come to a plan that is best for you and your family.
Both cfDNA and traditional screening tests have the possibility of a false positive or false negative result. A false positive result is when a test comes back high risk for a condition, such as trisomy 18, but the pregnancy does not actually have trisomy 18. On the other end, a false negative result is when a test comes back negative for a condition but the pregnancy actually has the condition.
Currently, cfDNA is not regulated by the FDA. It is also important to note that much of the information available regarding how reliable this test is comes from studies funded by the commercial labs and/or authored by individuals associated with one of the commercial labs.
cfDNA Testing: How do I decide?
The decision of whether or not to pursue prenatal genetic testing is up to you. Your doctor and genetic counselor are available to you with all of the information you need to make an informed decision that fits with your beliefs, values, needs, and personality. Your healthcare providers should also be there to support you in any decision you make regarding genetic screening in your pregnancy.
The following are some questions that may be helpful to consider if you are on the fence about whether to have cfDNA (or any other) prenatal screening:
- How would you feel if results indicated a higher risk for a genetic condition?
- Would you consider amniocentesis?
- If not, would you be ok waiting until the baby is born to know for sure if the condition is present if your prenatal cfDNA screening result returns high risk?
- Do you think this information would help you feel more prepared?
- Would you consider doing anything differently if you knew the baby had a genetic condition such as prepare, consider placing baby for adoption, or consider not continuing the pregnancy?
- Does more information with the possibility of uncertainty make you anxious?
- Some women prefer definitive answers and since prenatal cfDNA screening cannot provide that, they may choose to go straight to a more definitive test, such as chorionic villus sampling (CVS) or amniocentesis.
- Some women may feel comfortable with their chance of a genetic condition or are confident that, even if the baby did have a genetic condition, it wouldn’t alter their pregnancy plans. Or they may prefer not to face the decision of whether or not to have an amniocentesis if the prenatal cfDNA screening comes back high risk. In these cases some women may decide not to undergo any prenatal screening or diagnostic testing.
cfDNA Testing: Understanding the Results
Interpreting any prenatal genetic screening result can be challenging. Because of this, it may be helpful to meet with a specialist, such as a genetic counselor, who can review the specifics of your situation to help you understand what your cfDNA results mean for your pregnancy.
It is important to remember that cfDNA results, just like other screening results, do not give a ‘yes’ or ‘no’ answer to whether a pregnancy has a chromosome condition. cfDNA may tell you whether the pregnancy is at increased or decreased risk for the conditions that are being tested.
For tests that can provide you with a definitive ‘yes’ or ‘no’ answer, read more about prenatal diagnostic testing.
There are many labs that perform cfDNA testing, and each lab may report their results out in a slightly different way. Here are some examples:
- Positive (increased risk) or negative (decreased risk)
- Aneuploidy detected (increased risk) or no aneuploidy detected (decreased risk)
- Aneuploidy = extra or missing chromosome material was found
- High risk, intermediate risk, or low risk
Most labs will give you a separate result for each of the conditions they are screening for. For example, you may get a positive/high risk result for Down syndrome, but negative/low risk results for trisomy 18, trisomy 13, etc.
There are some additional things to keep in mind with regard to cfDNA screening results:
- How well the test works for Down syndrome, trisomy 18, trisomy 13, and other chromosome conditions varies widely. It is important to discuss your results with your provider to understand what they mean for you and your pregnancy.
- Not all labs screen for the same conditions.
- cfDNA screening can often tell if the baby is a boy or a girl. However, there is a small possibility that the test will predict fetal sex incorrectly.
- cfDNA screening does not test for all chromosome differences or birth defects.
- In some cases, cfDNA screening can find evidence of other health conditions that are present either in the baby or in the mother that were not expected.
- For example, cfDNA screening in rare cases has identified that a pregnant mom has cancer.
This is a basic overview of cfDNA screening results. It is important to meet with a specialist, such as your doctor or a genetic counselor, to understand what these results mean for you and your pregnancy.
cfDNA Testing: Understanding No-Call Results
With cfDNA testing, there is a possibility to get a no-call result. A no-call result means that the lab was not able to run the test, or that the test did not produce a result.
There are a few possible reasons that prenatal cfDNA screening may not provide a result. It could be due to poor DNA quality, or due to a problem with the shipping of the sample. Some labs look at something called fetal fraction. Fetal fraction represents the percentage of placental DNA that is floating around in mom’s blood.
Many women will get a result if they have the cfDNA screening repeated with a second blood draw. Some studies have indicated that receiving a no-call result may mean there is an increased risk for a chromosome condition, such as Down syndrome. Your doctor may offer you a diagnostic test, such as chorionic villus sampling (CVS) or amniocentesis to get more information. Diagnostic tests can provide more definitive information, but comes with a usually small risk for miscarriage.
cfDNA Testing: Calculating the Statistics
Understanding statistics can be challenging. Unfortunately, familiarity with some statistics are necessary to help you understand your genetic test results.
One statistical term that is important in prenatal genetics, particularly with cfDNA, is positive predictive value (PPV). PPV answers the question:
If my test comes back ‘high-risk’ or ‘abnormal’, what are the chances that someone in my exact position (same age, background risk, etc.) will have a baby with the condition that I tested positive for?
In other words, what are the chances that my positive test result is a true or actual positive?
The equation that is used to calculate PPV is:
Sensitivity X Prevalence / [Sensitivity X Prevalence + (1 – Specificity)(1 – Prevalence)]
Let’s break all of these terms down:
- Sensitivity measures the proportion of babies WITH a condition who are correctly identified. For example, if 100 babies have Down syndrome and the test detects 60 of the babies who have it, then the test would have a sensitivity of 60% for Down syndrome.
- For Down syndrome specifically, most labs quote a greater than 99% sensitivity. This means that for most people who take the test, there is over a 99% chance that if the baby has Down syndrome the test will come back ‘positive’ or ‘high-risk’. When a lab says there is a greater than 99% sensitivity, it does NOT mean that if you receive a ‘positive’ or ‘high-risk’ result there is a 99% chance that your baby has that condition.
- Specificity measures the proportion of babies WITHOUT a condition who are correctly identified. For example, if testing was done for 100 babies that do not have a chromosome condition, and 80 received a ‘normal’, ‘negative’, or ‘low-risk’ result, then the specificity of the test would be 80%. This would also mean that 20% of the babies would have a false positive result, meaning the result was ‘positive’ or ‘high-risk’, but the baby does not actually have the condition.
- For Down syndrome specifically, most labs quote a greater than 99% specificity. This means that if a pregnancy does NOT have Down syndrome, there is a greater than 99% chance that the test will come back ‘negative’ or ‘low-risk’.
- Prevalence is how common the condition is in the selected population. For example, chromosome conditions such as Down syndrome tend to happen more frequently as women get older. The chance for a 25-year-old woman to have a baby with Down syndrome is approximately 1 in 1250, while the chance for a 30-year-old woman is 1 in 840. A 35-year-old woman has a 1 in 356 chance to have a baby with Down syndrome.
Many labs do not provide a PPV calculation, but your genetic counselor or other provider should be able to help you calculate the PPV. You can also use this free PPV/NPV Calculator.
While most screening tests we talk about during pregnancy are designed to give us more information about the baby, genetic carrier screening is a test that gives us information about mom and dad.
As you may recall from Genetics 101, we have two copies of most genes: one we get from our mom and one we get from our dad. When we say that someone is a ‘carrier’ for a genetic condition, it means that there is a harmful change (called a pathogenic variant) in one of the copies of the gene that make it not work how it should. Because there is still one gene that is working properly, carriers generally do not show signs or symptoms of any health issues related to being a carrier.
Genetic carrier screening usually looks for conditions that are recessive, meaning that for someone to have the genetic condition both copies of the gene need to be not working. In most instances, if someone is a carrier for a recessive condition, they don’t have any medical or health problems, and may not have a family history of any genetic conditions.
Because being a carrier for a genetic condition does not cause any health problems in most cases, then what’s the point of testing for it? We are all probably carrier for five or more recessive genetic conditions. If two people who are carriers for the same genetic condition have a child, there is a 25% chance that they will have a child that has that genetic condition. This means there is a 1 in 4 chance that each parent will pass down their non-working copy of the gene to the pregnancy, and then the baby will have no working copies of that gene.
Carrier screening may also offer testing for X-linked recessive conditions. X-linked means that the gene is located on the X chromosome. To review from Genetics 101, women have two X chromosomes and men have one X and one Y chromosome (and thus only one copy of all of the genes on the X chromosome). If a male inherits a nonworking gene on his X chromosome, he usually will have that genetic condition because he does not have a second working copy of that gene.
If a woman, on the other hand, inherits a nonworking gene on one of her X chromosomes, she usually has a working copy of that gene on her other X chromosome. If a woman is a carrier for an X-linked recessive condition, she may have very mild or no symptoms at all. A woman who is a carrier for an X-linked recessive genetic condition would have a chance to pass that condition down to her children. Click here to see a list of commonly tested conditions.
How it Works
Genetic carrier screening can generally be done by either a blood or a saliva test. The blood or saliva is sent to the lab, where they can then pull out your DNA from either your blood cells or your cheek cells that are in your saliva. This DNA is what makes up your genes. Your genes are basically a string of letters that are an instruction for how to make a protein. Each protein has a very specific job to do in the body. Some proteins tell our body how tall we’re going to be, while others tell our eyes what color they will be.
Some proteins perform such important jobs in the body, that when we’re born without them, they can cause health problems, known as genetic conditions. If there is a harmful spelling error in the DNA, known as a pathogenic variant, it can change the instructions for that gene. If the instructions are changed, then that gene can make either an abnormal protein or no protein at all. Not having these proteins can lead to different health concerns. To learn about the different kinds of pathogenic variants, see sequencing and deletion/duplication testing.
Each lab offers screening for various genetic conditions. Most of the conditions that are offered for prenatal, or preconception carrier screening are passed down in a family in a pattern we call autosomal recessive, although there are some X-linked conditions that can be screened for.
Expanded Carrier Screening
Some medical providers offer expanded genetic carrier screening. Expanded carrier screening offers testing for many genetic conditions at one time. There are many viewpoints and ethical issues involved with expanded carrier screening, and like most things there are pros and cons.
Expanded carrier screening is available to all individuals, regardless of ethnicity, but your insurance may or may not cover this testing. Some of the conditions that expanded carrier screening looks for are severe, while others may be milder, or not even have any significant medical concerns associated with it. Some of these conditions have available treatments, while others don’t.
There is also a very small chance that you could find out that you actually have a genetic condition, depending on the genetic conditions that are screened for.
Labs that offer expanded carrier screening may or may not be as thorough as labs that do not focus on expanded carrier screening. For example, if you have a family history of a specific condition, expanded carrier screening may not be the best test for you.
Whether or not to move forward with expanded carrier screening can be a complicated decision, and it is important to discuss it with a medical provider, such as a genetic counselor, to fully understand what the test is able to tell you.
How to Decide
The choice of whether or not to undergo genetic carrier screening is yours. There may be some questions that would be helpful to consider before you make the decision on whether or not to pursue carrier screening:
- Would I like to know as much information as possible to plan and be prepared?
- Would having more information make me more anxious or nervous?
- If testing on my partner and I indicated an increased risk for a genetic condition, would I want to move forward with diagnostic testing (such as CVS or amniocentesis), that would be able to give a ‘yes’ or ‘no’ answer, but also carries a risk for miscarriage?
- If I knew that my baby had one of these conditions, would it affect my decision to continue the pregnancy? Would I consider alternative options, such as terminating the pregnancy, or placing the baby up for adoption?
This is not an inclusive list of things to consider but may be a good place to start. It is important to also discuss what sort of screening would be right for you with your doctor or genetic counselor.
Carrier Screening Results
My results were normal/negative. What does that mean?
When it comes to genetic carrier screening, a negative result significantly reduces, but does not eliminate, the chance to be a carrier of those genetic conditions. The chance to be a carrier for these genetic conditions after a negative genetic test is called the residual risk. The residual risk will vary depending on the laboratory that runs the test and the type of testing that was performed. Your provider or genetic counselor can help you determine what your residual risk is after having a negative genetic carrier screening test.
My results were positive, or said that I’m a carrier for a genetic condition. Now what?
To be a carrier for a genetic condition generally means that you have one working and one non-working copy of a gene. Most of these genes follow autosomal recessive inheritance patterns, which mean that someone would have to inherit two non-working copies of a gene in order to develop that genetic condition. For example, if someone inherits a non-working copy of the CFTR gene from both parents, it causes cystic fibrosis. However, if someone carries one non-working copy of a gene, they generally do not have any symptoms. However, they do have a higher chance to have a child that may have that genetic condition.
It is not uncommon to be a carrier for a genetic condition. We have over 20,000 genes, so everyone is likely a carrier for something. The next steps somewhat depend on what condition you were found to be a carrier for. If you were a carrier for a condition with autosomal recessive inheritance, the next step would be to consider carrier testing for your partner to see if they are also a carrier for the same genetic condition. If you and your partner are both found to be carriers for the same autosomal recessive genetic condition, there is a 25% chance for both parents to pass down the non-working copy of the gene, meaning that the baby would have that genetic condition.
If you are found to be a carrier for an X-linked condition, for women the chance to have an affected baby depends on whether the baby is a boy or a girl. If a woman is a carrier for an X-linked condition and is having a boy, there is a 50% chance to pass down the X chromosome with the nonworking gene, which would result in the baby having the genetic condition. There is also a 50% chance to pass down the X chromosome that has the working copy of the gene, which would result in a baby boy that does not have the genetic condition.
If the woman is a carrier for an X-linked condition and is having a girl, there is a 50% chance to pass down the X chromosome with the nonworking gene, which would mean that the baby girl would be a carrier for that genetic condition just like her mom. There is also a 50% chance to pass down the X chromosome that has the working copy of the gene, which would result in a baby girl that is not affected with the genetic condition and is also not a carrier for the genetic condition.
Both my partner and I are carriers of the same autosomal recessive genetic condition; what does this mean for our baby/potential baby and what are our options?
If you and your partner are both found to be carriers for the same genetic condition, there is a 25% (or 1 in 4) chance that the baby will inherit a non-working copy of the gene from both parents. That also means that there is a 50% chance that the baby will inherit one working and one non-working copy of the gene (which would make them carriers, just like their parents). There would also be a 25% chance that the baby will inherit a working copy of the gene from both parents.
Finding out that you and your partner are both carriers for the same autosomal recessive condition can be stressful. There are a couple of different options moving forward: either have prenatal diagnostic testing to find out of the baby is affected or wait until the baby is born and then revisit the option of genetic testing.
Prenatal diagnostic testing, such as chorionic villus sampling (CVS) and aminocentesis, comes with some risk (up to a 1 in 200 (0.5%) to 1 in 300 (0.03%) chance for a complication that could lead to a miscarriage from the procedure), but can provide a ‘yes’ or ‘no’ answer as to whether the baby is affected or not. For most genetic conditions, diagnostic testing can be done after the baby is born. The decision of whether to move forward with prenatal diagnostic testing is very personal and should be done after a talking through the risks, benefits, and limitations with your provider.
I found out that my partner and I are both carriers for the same recessive genetic condition and are concerned about having a child with a genetic condition. What are my options?
The answer to this question may depend on many things, including how severe the condition is, and what your personal thoughts are around having a child with a genetic condition. If you find that you and your partner are carriers for the same recessive genetic condition, there are several options:
- Move forward with pregnancy (or future pregnancy) as planned, and decline further genetic testing
- Move forward with pregnancy (or future pregnancy) and undergo diagnostic testing, such as CVS or amniocentesis, to find out during the pregnancy if the baby has the genetic condition. Some people want to pursue prenatal genetic diagnosis to be prepared for when the baby arrives, while other people want prenatal diagnosis because they would end a pregnancy that was affected. Other people would make plans for adoption if they found out the baby had a genetic condition. No matter the reason, it is important to discuss the risks, benefits, and limitations of diagnostic testing with your provider.
- Pursue in-vitro fertilization (IVF) with preimplantation genetic diagnosis (PGD). IVF with PGD involves fertilizing an embryo in a the lab, and then testing it to see if it has that genetic condition. You would then have the option to only transfer embryos that were not affected with the genetic condition. This process can be expensive, but some insurances may cover part or all of it.
- Consider using a sperm/egg donor
- Consider adoption
These options are not all easy, some of them cost a lot of money, and some of them may not fit with your personal beliefs and values. In terms of emotional support, it may be helpful to connect with another couple who has gone through this process; your provider or genetic counselor may be able to help facilitate this.
Prenatal Diagnostic Tests
Prenatal diagnostic tests are tests that are able, in most cases, to give you a definitive ‘yes’ or ‘no’ answer about whether a pregnancy is affected with a variety of chromosome and genetic conditions. While these tests are able to provide a lot of definitive information, there is a risk for miscarriage. The level of risk depends on which test is being done and the level of experience of the doctor performing the procedure. For tests that can still provide some information, but carry no risk for miscarriage, see Prenatal Screening Tests. It is also important to know that prenatal diagnostic tests are always optional. You never have to undergo any prenatal testing if you do not wish to. Anyone who is having prenatal testing should have access to all of the necessary information to make an informed decision about what is right for them and their family.
Amniocentesis is a procedure that can be done during the second trimester of pregnancy (usually between 15 and 22 weeks). Amniocentesis involves getting some of the fluid from around the baby so that the lab can test for chromosome and genetic conditions.
Because amniocentesis is an invasive procedure, there is a risk of less than 1 in 200 (or 0.5%) for complications that can lead to a miscarriage. That also means that more than 99.5% of women who have an amniocentesis will not have a procedure-related miscarriage.
There are many different types of testing that can be done on amniocentesis samples, including traditional chromosome analysis (karyotype), FISH, chromosomal microarray, and familial mutation testing. All of this testing is considered to be diagnostic testing for chromosome and genetic conditions, meaning it can typically provide a ‘yes’ or ‘no’ answer, as opposed to screening tests which only tell you the chances or odds.
Amniocentesis can screen for many things, but cannot screen for all medical conditions or birth defects.
How does the procedure work?
An amniocentesis procedure can be done in the doctor’s office by a high-risk pregnancy doctor called a perinatologist. Amniocentesis is an outpatient procedure, meaning you can usually go home soon afterward. Most women find that it can be uncomfortable and many compare it to having intense menstrual cramps, but generally do not experience considerable pain beyond that.
The point of the procedure is usually to obtain fluid from around the pregnancy to send to the lab for testing. The lab can look at cells from the pregnancy that are floating in the fluid, and inside those cells at the chromosomes. To learn more about chromosomes, visit our Genetics 101 page.
To start with, generally an ultrasound technician will perform an ultrasound to allow the doctor to find a pocket of fluid that is furthest away from the pregnancy. With the ultrasound on the entire time, the doctor inserts a small needle into the abdomen (not through the belly button), through the uterus, and into the sac that the pregnancy is in. Once the needle is in the sac, a syringe is attached to the end and fluid is drawn out and sent to the lab.
Because the uterus is a muscle, some women may notice intense cramping during and immediately after the procedure. Some people have concerns about the needle poking the baby; the ultrasound is constantly monitoring where the baby is to ensure that they do not get close to the needle. Most doctors also use a plastic catheter to remove the fluid after they have gotten into the sac, so if the baby were to brush up against the tube it would not cause any harm.
Because amniocentesis is an invasive procedure, there is an approximated less than 1 in 200 (0.5%) chance that it may cause complications that could lead to a miscarriage. This also means that over 99.5% of women who undergo amniocentesis testing do not have a procedure-related miscarriage. To help limit the risk for complications, your doctor may give you special instructions to limit strenuous physical activity for the few days following your procedure.
Depending on the type of testing that is performed, amniocentesis can look for chromosome conditions, such as Down syndrome, or other genetic conditions, but cannot rule out all birth defects or genetic conditions.
How do I decide?
The decision of whether or not to have a diagnostic test, such as amniocentesis, can be a difficult decision to make. Your genetic counselor can help answer any questions, and help you come to the decision that is best for your personality, beliefs, and values. Some things that may be helpful to think about if you are thinking about undergoing an amniocentesis procedure are:
- Do I feel like I need ‘yes’ or ‘no’ answers to be able to enjoy the rest of my pregnancy?
- If I feel like I need a ‘yes’ or ‘no’ answer, and I OK with the small risk for miscarriage that is associated with these procedures (generally thought to be less than 1 in 200, or 0.5%)?
- Would I feel OK waiting until the baby arrives to know for sure if they have one of these conditions?
- If I knew that the pregnancy had a chromosome condition, would it affect my decision on whether or not to continue the pregnancy?
- If I knew that the pregnancy had a chromosome condition, would it affect my decision on what to do after the baby arrives (such as adoption)?
- Are there other trusted people in your life that you can turn to for advice and perspective (i.e. religious leader, family member, friend)?
If someone is still having difficulty deciding whether or not they want a diagnostic procedure, it may be helpful to think of the various outcomes. If someone would end a pregnancy if it was found to have a chromosome condition, then the two outcomes that would be the least favorable would be either:
- Not having a diagnostic procedure, such as amniocentesis, and then finding out later that the pregnancy had a chromosome condition that could have been diagnosed early in pregnancy, OR
- Having a diagnostic procedure, such as amniocentesis, and then having a subsequent miscarriage, and finding out that the pregnancy did not have a chromosome condition.
Either of these ‘worst case’ scenarios could be difficult to handle, but if you were faced with them both, which seems like the more difficult option?
These questions are not meant to be the be-all end-all of things to consider when deciding whether or not to have an amniocentesis, but they can be a good place to start.
What results can I expect to get?
The type of results may vary depending on the type of testing that is done on the amniocentesis sample. The four main types of testing that can be done on amniocentesis samples are: traditional chromosome analysis (also called a karyotype), chromosomal microarray, familial mutation testing, and fluorescent in-situ hybridization (FISH). Click on each of these to learn more about the different types of results that you can get with each type of testing.
My results are abnormal. Now what?
This can be difficult news to hear, and everyone handles situations like this in different ways. It is often helpful to talk with your doctor or genetic counselor to make sure any questions that you have are answered, particularly if the results are complex or confusing. Even if you are familiar with the condition, it can be helpful to talk things through with your provider, or to get any up-to-date information, as things in the medical field change rapidly.
What comes next is a very personal and often challenging decision to make. There are generally three ways to move forward once you find out that the baby has a chromosomal or genetic condition:
- Continue the pregnancy and make preparations (both mentally and medically) for when the baby gets here
- Continue the pregnancy and place the baby up for adoption. There are many programs available that can help find a family for a baby with special needs.
- Do not continue the pregnancy
Your doctor or genetic counselors can help to answer any questions you may have about your options, and may have other resources that may be helpful.
Chorionic villus sampling, or CVS, is a procedure that can be done during the first trimester of pregnancy (usually between 11 and 14 weeks). CVS involves getting some of the tissue from the placenta (the chorionic villi) so that the lab can test for chromosome and genetic conditions.
Because CVS is an invasive procedure, there is a risk of less than 1 in 200 (or 0.5%) for complications that can lead to a miscarriage. That also means that more than 99.5% of women who have a CVS will not have a procedure-related miscarriage. Because of this risk for complications, diagnostic tests like CVS are always optional, and never anything that you would have to do.
There are many different types of testing that can be done on CVS samples, including traditional chromosome analysis (karyotype), FISH, chromosomal microarray, and familial gene testing. All of this testing is considered to be diagnostic testing for chromosome and genetic conditions, meaning it can typically provide a ‘yes’ or ‘no’ answer, as opposed to screening tests which only tell you the chances or odds.
CVS can screen for many things, but cannot screen for all medical conditions or birth defects, including open neural tube defects.
How does the procedure work?
A CVS procedure can be done in the doctor’s office by a high-risk pregnancy doctor called a perinatologist. CVS is an outpatient procedure, meaning you can usually go home soon afterward. Most women find that it can be uncomfortable and many compare it to having intense menstrual cramps, but generally do not experience considerable pain beyond that.
The point of the procedure is usually to get tissue from the placenta to send to the lab for testing. The lab can look at the cells from that tissue, and inside those cells at the chromosomes. Approximately 98-99% of the time, the baby and the placenta have the same chromosomes. To learn more about chromosomes, visit our Genetics 101 page.
Approximately 1-2% of the time, the placenta’s chromosomes may be different from the baby’s chromosomes; this is called confined placental mosaicism (or CPM) – see below for more information about CPM. If the test results show extra or missing chromosomes (called aneuploidy) and the lab is able to confirm that the aneuploidy is caused by CPM, then the most likely thing is that cells with aneuploidy are just in the placenta and not in the baby. The only way to tell this for sure before the baby is born would be to do another test called an amniocentesis.
There are two possible ways that the CVS may be done:
- A catheter can be used to reach the placenta vaginally
- A small needle can be inserted into the abdomen to reach the placenta
Whether a CVS is done via the abdomen or vaginally is dependent on many factors, including where the placenta is located and the doctor who is performing the procedure. The procedure generally takes less than a couple minutes from start to finish.
Because CVS is an invasive procedure, there is a less than 1 in 200 (0.5%) chance that it may cause complications that could lead to a miscarriage. This also means that over 99.5% of women who undergo CVS testing do not have a procedure-related miscarriage. To help limit the risk for complications, your doctor may give you special instructions to limit strenuous physical activity for the few days following your procedure. Because of this risk for complications, diagnostic tests like CVS are always optional, and never anything that you would have to do.
Depending on the type of testing that is performed, CVS can look for chromosome conditions, such as Down syndrome, or other genetic conditions, but cannot rule out all birth defects or genetic conditions.
What is Confined Placental Mosaicism (CPM)?
Because the placenta and the baby come from the same cells, they have exactly the same amount of chromosomes most (98-99%) of the time. In approximately 1-2% of all pregnancies, the placenta can actually have a different chromosome makeup from the baby. This is called confined placental mosaicism (CPM).
When it comes to genetic testing, CPM is important to remember particularly when it comes to CVS procedures. If someone has a CVS and the results show an extra or missing chromosome, we know that the most likely thing is that the pregnancy also has that extra or missing chromosome. However, 1-2% of the time, that extra or missing chromosome is only found in the placenta and not in the pregnancy.
The exact reason for this difference is unknown, but it is thought that after the placenta and the baby separate, some sort of error happens during cell division in the placenta, which causes some of the cells to have an extra or missing chromosome.
If CPM is suspected on a CVS result, it may be recommended to perform an amniocentesis procedure to confirm. Because the cells obtained from an amniocentesis are from directly around the baby, it is more accurate in determining whether the pregnancy actually has the extra or missing chromosome.
How do I decide?
The decision of whether or not to have a diagnostic test, such as a CVS, can be a difficult decision to make. Your genetic counselor can help answer any questions, and help you come to the decision that is best for your personality, beliefs, and values. Some things that may be helpful to think about if you are thinking about undergoing a CVS procedure are:
- Do I feel like I need ‘yes’ or ‘no’ answers to be able to enjoy the rest of my pregnancy?
- If I feel like I need a ‘yes’ or ‘no’ answer, and I OK with the small risk for miscarriage that is associated with these procedures (generally thought to be less than 1 in 200, or 0.5%)?
- Would I feel OK waiting until the baby arrives to know for sure if they have one of these conditions?
- If I knew that the pregnancy had a chromosome condition, would it affect my decision on whether or not to continue the pregnancy?
- If I knew that the pregnancy had a chromosome condition, would it affect my decision on what to do after the baby arrives (such as adoption)?
- Are there other trusted people in your life that you can turn to for advice and perspective (i.e. religious leader, family member, friend)?
If someone is still having difficulty deciding whether or not they want a
diagnostic procedure, it may be helpful to think of the various outcomes. If someone would end a pregnancy if it was found to have a chromosome condition, then the two outcomes that would be the least favorable would be either:
- Not having a diagnostic procedure, such as a CVS, and then finding out later that the pregnancy had a chromosome condition that could have been diagnosed early in pregnancy, OR
- Having a diagnostic procedure, such as a CVS, and then having a subsequent miscarriage, and finding out that the pregnancy did not have a chromosome condition
Either of these ‘worst case’ scenarios could be difficult to handle, but if you were faced with them both, which seems like the more difficult option? These questions are not meant to be an inclusive list of things to consider when deciding whether or not to have a CVS, but they can be a good place to start.
What results can I expect to get?
The type of results may vary depending on the type of testing that is done on the CVS sample. The four main types of testing that can be done on CVS samples are: traditional chromosome analysis (also called a karyotype), chromosomal microarray, familial gene testing, and fluorescent in-situ hybridization (FISH). Click on each of these to learn more about the different types of results that you can get with each type of testing.
My results are abnormal. Now what?
This can be difficult news to hear, and everyone handles situations like this in different ways. It is often helpful to talk with your doctor or genetic counselor to make sure any questions that you have are answered, particularly if the results are complex or confusing. Even if you are familiar with the condition, it can be helpful to talk things through with your provider, or to get any up-to-date information, as things in the medical field change rapidly.
What comes next is a very personal and often challenging decision to make. There are generally three ways to move forward once you find out that a pregnancy has a chromosomal or genetic condition:
- Continue the pregnancy and make preparations (both mentally and medically) for when the baby gets here.
- Continue the pregnancy and place the baby up for adoption. There are many programs available that can help find a family for a baby with special needs.
- Do not continue the pregnancy.
It is important to remember that there is no ‘right’ or ‘wrong’ thing to do in this situation; only what is the right choice for you and your family. Your doctor or genetic counselor can help to answer any questions you may have about your options, and may have other resources that may be helpful.
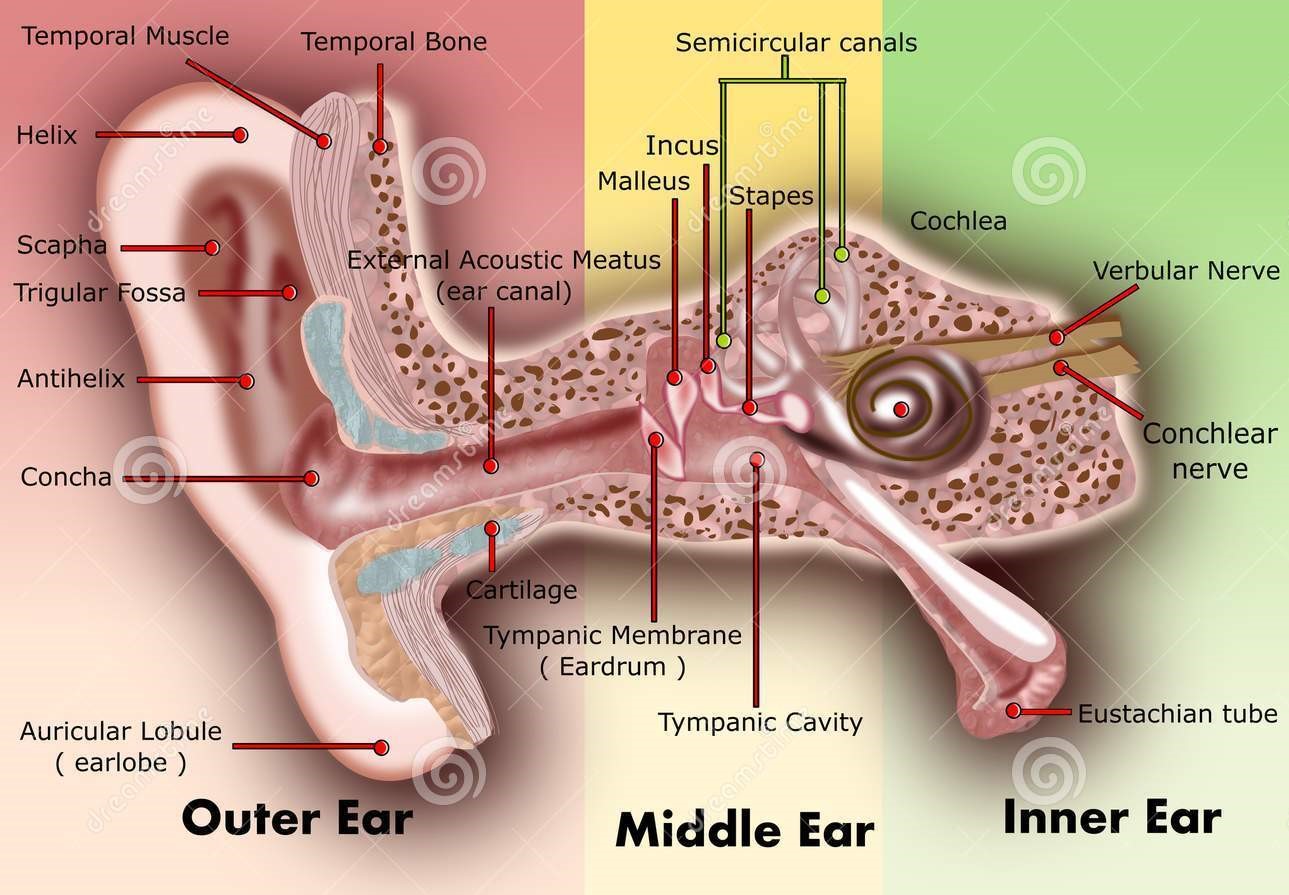
FISH, or fluorescence in situ hybridization, is a test that can be done on the cells from a chorionic villus sampling (CVS) or amniocentesis procedure. FISH can provide rapid results (usually within 48 to 72 hours) for limited chromosome conditions. Most labs offer FISH testing on CVS or amniocentesis cells for chromosomes 21, 18, 13, and the sex chromosomes (X and Y). To learn more about chromosomes, visit our Genetics 101 page.
Each of our chromosomes have sequences of letters, our DNA, that are unique to them. Our DNA is made up four different letters, called nucleotides: A, C, G, and T. These nucleotides work in pairs – A always pairs with T, and G always pairs with C.
Every chromosome has a pattern of these letters that are different than all of the other chromosomes, which is how we can identify them. Once we know what that pattern is, the lab can make a probe (which is a small piece of DNA that is made by the lab) that can find and attach to the matching sequence.
For example, if we know chromosome 18 has the sequence ACG TAT CGC, then the lab would make a probe that has the sequence TGC ATA GCG (remember T matches with A, G matches with C, etc). Once this probe is made, the lab can put it in with the cells from the CVS or amniocentesis procedure. When the probe finds it’s matching chromosome, it attaches and then lights up (like a fluorescent light!).
FISH results on CVS and amniocentesis will generally tell you how many of chromosomes 21, 18, 13, X, and Y the lab saw. We generally have two of each chromosome, so if the FISH test shows that three chromosome 21s lit up, that means that there is a high chance that the pregnancy has three number 21 chromosomes, which is what causes Down syndrome.
Because the FISH testing also looks at the X and Y chromosomes, it will also be able to tell whether the pregnancy is genetically male or female. So, if you don’t want any spoilers, make sure your doctor or genetic counselor know ahead of time!
Although FISH is thought to be a reliable preliminary test, it is most often recommended to wait until the full genetic testing results have come back before making any permanent decisions about the pregnancy.
Prenatal Carrier Screening Tests
The field of prenatal screening has rapidly evolved over the last few years, and will likely continue to change in the future. There are many different types of screening tests available during pregnancy. It is important to know that screening tests will only tell you about the chance of a medical condition. Screening tests will not give you a ‘yes’ or ‘no’ answer. It is also important to know that prenatal screening tests are always optional. You never have to undergo any prenatal testing if you do not wish to. Anyone who is having prenatal testing should have access to all of the necessary information to make an informed decision about what is right for them and their family.
There are a number of genetic disorders that occur in people from any ethnic background, but are more common in people who have Ashkenazi Jewish ancestry. These conditions can vary from little to no medical complications to severe and life-limiting illness
Screening for the following conditions should be offered to all individuals (pregnant or planning a pregnancy) with Ashkenazi Jewish ancestry, according to the American College of Medical Genetics and Genomics (ACMG):
- Familial dysautonomia
- Tay-Sachs disease
- Canavan disease
- Fanconi anemia group C
- Niemann-Pick type A
- Bloom syndrome
- Mucolipidosis IV
- Gaucher disease, type 1
Your doctor or genetic counselor can help you determine the best screening approach if you have Ashkenazi Jewish ancestry.
Cystic fibrosis (CF) is an inherited genetic condition that mainly affects the lungs. However, people with CF can also have issues that affect their digestive system, reproductive system, and other organs in the body. Mucus glands typically produces mucus to cover and protect many different organs, including the lungs. The mucus glands in people who have CF are not able to properly regulate themselves. The mucus glands in people with cystic fibrosis produce more mucus than they should, and it is abnormally thick and sticky. This thick and sticky mucus leads to breathing problems and bacterial infections in the lungs, which can lead to permanent lung damage. Because the digestive system also requires mucus, some individuals with CF will also have thick and sticky mucus that will buildup in the pancreas, causing digestive problems such as diarrhea, malnutrition, poor growth, and weight loss. Because mucus also helps the pancreas to function properly, some individuals may even develop diabetes because of poor insulin production (insulin is made in the pancreas). Most individuals who have CF are found through routine newborn screening.
There are forms of CF (called non-classic) that are not associated with the potentially severe health concerns that people with classical CF have. In men, mucus lines their vas deferens, which are the tubes that carry sperm from the testicles out of the body. Some men with non-classic CF can have a condition called congenital absence of the vas deferens (CBAVD), which is when the vas deferens are blocked by mucus and do not develop properly. Men with CBAVD are infertile without undergoing fertility treatments, but they often will not develop the lung or gastrointestinal symptoms that people with classic CF have.
Causes of Cystic Fibrosis
We have over 20,000 different genes in the body. These genes are like instruction manuals for how to build a protein, and each protein has an important function that helps to keep our body working how it should. The CFTR gene makes a protein called the cystic fibrosis transmembrane conductance regulator (CFTR) protein. The CFTR protein works as a channel to move substances called chloride ions in and out of our cells. Moving these chloride ions in and out of our cells is what helps to control the amount of water that is in our tissues. This balance is important for our body to make normal, thin mucus. The CFTR protein also works to move substances called sodium ions in and out of cells, which helps some organs in our body (like the lungs and pancreas) to function how they should.
If someone has a harmful change (called a pathogenic variant) in both of their CFTR genes (the one they got from their mom and the one they got from their dad), then their body is not going to make enough of the CFTR protein. If the body does not have enough CFTR protein, then the cells are not able to move chloride and sodium ions in and out of the cells like they should. This is what leads to the symptoms we associate with CF.
CF is inherited in an autosomal recessive pattern. This means an individual who has CF has inherited two non-working copies of the CFTR gene; the one they inherited from their mom is not working and the one they inherited from their dad is not working. In the case of autosomal recessive conditions, if you inherit one working CFTR gene from a parent and one non-working CFTR gene from a parent, you are called a ‘carrier’ for CF. Carriers do not have CF, and typically do not have signs or features of CF.
How common CF is depends on someone’s ethnic background. It is estimated that CF affects approximately 1 in 2500 to 1 in 3500 newborns that are white, 1 in 17,000 newborns that are black, and 1 in 31,000 newborns that are of Asian descent.
Genetic Testing for Cystic Fibrosis
Genetic testing for pathogenic variants in the CFTR is currently available, but there are a couple different ways to approach testing:
- Single site analysis: Testing specific to a known pathogenic variant in the family
- Full gene sequencing and rearrangement analysis: Comprehensive testing to search for all currently detectable pathogenic variants in the gene
Diagnosing Cystic Fibrosis
Many infants with cystic fibrosis will be found through routine newborn screening. Further testing to confirm if a baby has CF can be done either through genetic testing, or sweat testing. The sweat test involves using a small amount of electrical stimulation (not painful but may cause some tingling) to get the body to produce sweat. That sweat is gathered on a paper and sent to the lab, where they measure the amount of a substance called chloride that is in the sweat. If there are larger amounts of chloride in the sweat than expected, that can lead to a diagnosis of CF.
Treatment/Management for Cystic Fibrosis
Management of CF is a lifelong process that includes taking steps to prevent lung problems and improve weight gain. The majority of individuals with CF will require medications that will make their mucus thinner and easier to cough up, other medications to open the airway to make breathing easier, and antibiotics for frequent lung infections. In addition, some individuals affected will need Airway Clearance Therapies (ACT), which help them clear sticky, thick mucus through coughing. Those individuals with digestive system problems may require pancreatic enzymes to help digest food properly. Individuals with malnutrition may also need a high-calorie diet and vitamin supplements for healthy growth and development. With advances in treatment and appropriate management, individuals with classic cystic fibrosis typically live into their late thirties, or longer. Individuals with CBAVD may conceive children through assisted reproductive technologies.
Fragile X syndrome (FXS) is an inherited condition and one of the most common causes of inherited intellectual disability (1-2% of all ID) and autism spectrum disorders (2-3% of all ASD). Individuals with FXS commonly have:
- developmental delays, including
- speech delays
- cognitive/intellectual delays
- social/behavioral problems (tantrums, ADHD/ADD, hand flapping, acting and speaking without thinking, and poor eye contact)
- autism spectrum disorder
- seizures
- flexible joints
- scoliosis
- flat feet
- macroorchidism (abnormally large testicles)
- heart issues (particularly mitral valve prolapse or aortic root dilation)
- hypotonia (decreased muscle tone)
- unique facial features, including:
- a longer face
- larger ears
- a prominent forehead, and/or
- a prominent jaw.
It is more common for males to show symptoms than females, and the symptoms in males tend to be more severe. FXS occurs in about 1 out of every 4000 males and 1 out of every 8000 females.
Causes of Fragile X Syndrome
FXS is caused by a repeat expansion of the FMR1 gene, and is inherited in an X-linked pattern. The FMR1 gene makes a protein called FMRP. One job this protein has is in the brain; it helps develop connections between nerve cells (called synapses). These connections are vital to making sure your brain can function how it’s supposed to. The synapses in your brain are constantly changing in response to experiences that you have, and FMRP is thought to help regulate the evolution of these connections. This is particularly important for learning and memory.
The FMR1 gene has a section where three letters (CGG, called a trinucleotide repeat) are repeated over and over. Everyone has these repeats in their FMR1 gene, but how many repeats the gene has is important. Most people have between 5 and 40 CGG repeats in their FMR1 gene. People who have FXS have over 200 CGG repeats (called a mutation). Having too many of these CGG repeats causes the FMRP protein that the gene makes to not work how it’s supposed to. Not having enough of this working FMRP protein is what leads to the signs and symptoms of FXS.
The FMR1 gene is located on the X chromosome. Women have two X chromosomes and males only have one (they have a Y chromosome instead of a second X chromosome), which is why it is more common to see males affected with FXS than females (females have a back-up copy of the FMR1 gene, while males don’t). Females are also usually less severely affected than males.
The number of CGG repeats that someone’s FMR1 gene has can increase (called an expansion) when it is passed down. For example, a woman could have one of her X chromosomes that has an FMR1 gene with 80 CGG repeats, but then when she passes it down to her daughter, it expands to 120 CGG repeats. It is less common for the expansion to happen when the FMR1 gene is passed down from someone’s father. If a woman has an FMR1 gene with over 90 CGG repeats, there is about a 94% chance that if she passes that copy of her FMR1 gene down that it will expand to over 200 CGG repeats. This could lead to her having a son with FXS or a daughter who is a carrier for FXS (and may also have symptoms).
The AGG of it all...
Some studies have shown that having what are called ‘AGG interruptions’ can decrease the chance that someone’s FMR1 gene will expand when it’s passed down. In addition of CGG repeats, the FMR1 gene also can contain the trinucleotide AGG within the CGG repeats (thus ‘interrupting’ the string of CGG repeats). Having these AGG interruptions may make the FMRP protein that the gene makes more stable and less likely to expand if it’s passed down. Currently, the impact of AGG repeats on the chance for FMR1 gene expansion are investigational and not supported by any medical guidelines, as more research is needed.
Premutation and Intermediate...
The smallest number of repeats that has expanded to a mutation (200 CGG repeats) is 56. Because of this, if someone has an FMR1 gene with between 55 and 200 CGG repeats, they are called a premutation carrier. FMR1 genes that have between 45 and 55 CGG repeats are called intermediate.
The FMR1 gene in premutation carriers is still able to make FMRP protein, so they are not at risk to have FXS. However, individuals who have a pre-mutation can be at a higher risk for Fragile X-Associated Tremor/Ataxia Syndrome (FXTAS). Women who are premutation carriers are also at increased risk for FMR1-Related Primary Ovarian Insufficiency (FXPOI).
FXTAS is a condition characterized by tremors (trembling or shaking) and ataxia (movement, coordination, and balance difficulties). Individuals with FXTAS can also have problems with involuntary body functions (such as bladder and bowel movement control) as well as mental health issues (depression, anxiety, mood changes, etc). Average age of onset of symptoms for FXTAS is typically between 60 to 65 years of age. Males also have a higher risk for FXTAS than women (40% compared to 16-20% for women), and tend to be more severely affected.
FXPOI is characterized by a woman going through menopause (stopping of periods) before the age of 40.. Women in the general population have about a 1% risk for primary ovarian insufficiency, while women who carry a pre-mutation have about a 20% risk. Women with FXPOI go through menopause an average of five years earlier than women without FXPOI.
Individuals who have an intermediate FMR1 gene (45-55 CGG repeats) are not considered to be carriers, and not at risk for FXTAS or FXPOI. There have been some studies that suggest a potential increased risk for developmental or behavioral disorders, but this remains uncertain and is likely unrelated.
Diagnosing Fragile X Syndrome
The diagnosis of FXS is made through genetic testing that looks at the number of CGG repeats that are in the FMR1 gene. If a male has over 200 CGG repeats in his FMR1 gene, then he has a diagnosis of FXS. If a female has over 200 CGG repeats in one of her FMR1 genes, then she is a carrier for FXS and may experience some symptoms. If a female has over 200 CGG repeats on BOTH of her FMR1 genes, then she has a diagnosis of FXS. FXTAS and FXPOI are diagnosed by a combination of clinical symptoms and having between 55-200 CGG repeats in their FMR1 gene
Genetic Testing for Fragile X Syndrome
Any individual who has intellectual/cognitive disability or developmental delay (where the cause is unknown) should consider testing for FXS. People with learning disabilities, social/behavioral problems, or autism spectrum disorder should also consider seeing a medical professional to evaluate for possible FXS. Individuals with personal or family histories of FXS-related health issues (late onset tremor or ataxia of unknown cause, early menopause) may also wish to consider fragile X syndrome screening.
Treatment/Management for Fragile X Syndrome
Treatment for fragile X syndrome is primarily focused on treatment of health issues that arise, and should be tailored to each individual. Some common treatments for individuals with FXS include:
- Therapy
- Behavioral intervention
- Medications
- Speech and language therapy
- Occupational therapy
- Individualized educational support
- Early intervention
Hemoglobin diseases, called hemoglobinopathies, are a group of disorders characterized by abnormal or decreased production of hemoglobin, the molecule that carries oxygen throughout the body. The severity of these diseases ranges from mild to severe, depending on the type of hemoglobin defect.
The American College of Obstetricians and Gynecologists recommends that screening for hemoglobinopathies be offered to couples where at least one of the partners has an ethnic background that includes any of the following:
- African American
- Mediterranean
- Southeast Asia
There are several different types of hemoglobinopathies, but the most common ones are thought to be:
- alpha thalassemia (see information below)
- beta thalassemia
- sickle cell anemia (see information below)
Alpha Thalassemia
Alpha thalassemia is a genetic disorder called a hemoglobinopathy, or an inherited type of anemia. People who have alpha thalassemia make red blood cells that are not able to carry oxygen as well throughout the body, which can lead to anemia. This chronic anemia can include pale skin, fatigue, and weakness. In more severe cases, people with alpha thalassemia can also develop jaundice (yellowing of the eyes and skin), heart defects, and an enlarged liver and spleen (called hepatosplenomegaly).
Alpha thalassemia is more common in people of African, Southeast Asian, Chinese, Middle Eastern, and Mediterranean ancestries.
Causes of Alpha Thalassemia
We have over 20,000 different genes in the body. These genes are like instruction manuals for how to build a protein, and each protein has an important function that helps to keep our body working how it should. The HBA1 and HBA2 genes make a protein called alpha-globin. Two of the alpha-globin proteins combine with two other proteins called beta-globins (which are made by the HBB gene) to make a normal red blood cell.
Most people have four copies of the genes that make the alpha-globin protein: two copies of the HBA1 gene (one from each parent), and two copies of the HBA2 gene (one from each parent). Having all four copies of these genes is symbolized by writing αα/αα. Whether or not someone has alpha thalassemia depends on how many working copies of the alpha-globin gene they have (if someone has a missing or nonworking alpha-globin gene, it is most frequently caused by a deletion):
- Silent alpha thalassemia carrier (also referred to as -α/αα): When there is one missing alpha-globin gene. Silent alpha thalassemia carriers do not usually have signs or symptoms of alpha thalassemia because their three working alpha-globin genes are enough to make up for the one that is missing.
- Alpha thalassemia carrier, or alpha thalassemia trait (also referred to as –/αα or -α/-α): When there are two missing alpha-globin genes. The two missing genes can be on the same chromosome (called the cis position, or –/αα) or there can be one missing gene on each chromosome (called the trans position, or -α/-α). Whether someone is more likely to be a cis or trans carrier for alpha thalassemia can depend on their ethnic background. Some people who have alpha thalassemia trait may have red blood cells that are smaller than normal (microcytosis). Most people who are carriers for alpha thalassemia have no symptoms, but some may experience anemia.
- Alpha thalassemia, or hemoglobin H disease (also referred to as –/-α): When there are three missing alpha-globin genes. This form of alpha thalassemia is extremely variable and can range from mild to moderate anemia. Some individuals with hemoglobin H disease will have no symptoms, while others may experience yellowing of the eyes and skin (jaundice), bone changes (overgrowth of the upper jaw and a prominent forehead), developmental delays, and gallstones, in addition to other health concerns.
- Alpha thalassemia major, or hemoglobin Bart disease (also referred to as –/–): When all four alpha-globin genes are missing. This is the most severe form of alpha thalassemia, and is characterized by excess fluid build up in the body before birth (hydrops fetalis). Unfortunately, most babies with hemoglobin Bart disease are stillborn or die shortly after birth. Alpha thalassemia major can also include severe anemia, an enlarged liver and spleen (hepatosplenomegaly), and defects of the heart, urinary system, and genitalia.
Alpha thalassemia is inherited in a complex autosomal recessive inheritance pattern, and potential outcomes depend on the parent’s specific test results (how many copies of the alpha globin gene do the parents have on each of their chromosomes).
Prenatal testing and/or pre-implantation genetic testing options are available for couples who are identified to be at an increased chance to have a baby with alpha thalassemia.
How common alpha thalassemia is depends on the part of the world someone’s family is from. People whose families are from Northern Europe and North America have about a 1 in 1,000,000 chance to have alpha thalassemia. Approximately 4-20 out of every 1000 people whose families are from the Middle East, Southeast Asia, and certain Mediterranean countries have hemoglobin H disease. Exact numbers for how common alpha thalassemia is may not be completely accurate, as many people can have mild or no symptoms and may not even know that they have it.
Treatment/Management for Alpha Thalassemia
Medical management for alpha thalassemia depends on the severity of symptoms that the person is experiencing. Because people who are silent carriers or those who have alpha thalassemia trait most frequently do not have any health symptoms related to this, no treatment is necessary.
Treatment for hemoglobin H disease may include taking folic acid supplements, blood transfusions (as needed), removal of iron (called iron chelation therapy) due to iron overload, and surgical removal of the spleen (splenectomy). Rarely, a bone marrow transplant (also called a stem cell transplant) may aid in the survival of a child with hemoglobin Bart syndrome. Bone marrow transplants carry many risks, so they are generally only considered in the most severe cases of alpha thalassemia.
Sickle Cell Anemia
Sickle cell disease is a genetic disorder called a hemoglobinopathy, or an inherited type of anemia. Most people’s red blood cells are a round shape, like a doughnut, and they deliver oxygen throughout our bodies to keep everything working correctly. People who have sickle cell anemia have red blood cells that are shaped like a sickle, or a banana. Red blood cells that are this shape can not carry as much oxygen around the body as normal red blood cells. Red blood cells that have the sickle shape break down and die more quickly than they should, which results in anemia. Anemia can cause shortness of breath, fatigue, pale complexion, and delayed growth and development in children.
Individuals with sickle cell anemia generally show some symptoms early in childhood, although the severity can vary greatly between people. Some other common health issues people with sickle cell are anemia (low number of red blood cells), swelling of the hands and feet, high blood pressure, heart failure, organ damage, and infections.
Because of their shape, there is also a chance that these sickled blood cells can get stuck in veins or arteries and lead to a blockage. This blockage prevents blood from flowing to part of the body and can cause a sickle cell pain crisis, which is when there is severe pain because a part of the body is not getting the necessary blood flow. This pain can be throbbing, sharp, dull, or stabbing, and can last anywhere from several hours to several days. This lack of oxygen can also cause organ damage, stroke, cognitive delays, blindness, ulcers, and gallstones.
Causes of Sickle Cell Anemia
We have over 20,000 different genes in the body. These genes are like instruction manuals for how to build a protein, and each protein has an important function that helps to keep our body working how it should. The HBB gene makes a protein called beta-globin. Two of the beta-globin proteins combine with two proteins called alpha-globins (which are made by the HBA1 and HBA2 genes) to make a normal red blood cell.
People who have sickle cell anemia have a specific change (called a pathogenic variant) in both of their HBB genes that changes the beta-globin protein. That altered protein is what gives the red blood cell it’s sickle shape (called hemoglobin S).
In more rare instances, someone can inherit the specific variant in their HBB gene for sickle cell from one parent, and then can inherit a different pathogenic variant in the HBB gene they get from their other parent. It is also possible for someone to inherit the sickle cell HBB gene variant from one parent, and then inherit a pathogenic variant in the genes that make the alpha-globin proteins (HBA1 and HBA2). These different combinations are overall thought to be rare.
Sickle cell anemia is inherited in an autosomal recessive pattern. This means an individual who has the sickle cell anemia has inherited two copies of the HBB gene (one from each parent) that both have the sickle cell variant. If someone has one normally functioning HBB gene, and one HBB gene that has the sickle cell variant in it, they are called a ‘carrier’ for sickle cell anemia (also called sickle cell trait).
It is estimated that approximately 8%-10% of African Americans are carriers for sickle cell anemia. Some research has shown that as many as 1 in 2 to 1 in 3 individuals from parts of sub-Saharan Africa have sickle cell trait. It is also thought to be more common in people with Hispanic, South Asian, Southern European, and Middle Eastern populations.
People with sickle cell trait do not have sickle cell anemia, and most do not have any direct health consequences because of it. However, in rare extreme conditions, people with sickle cell trait can develop some of the symptoms of sickle cell anemia. These situations include an increase in atmospheric pressure (can happen with activities such as scuba diving), low oxygen levels (can happen during strenuous physical activity), high altitudes, and dehydration.
Treatment/Management for Sickle Cell Anemia
Much of the treatment for sickle cell anemia surrounds avoiding circumstances that increase the chance for complications and having a plan to manage complications as they arise. It is recommended for people with sickle cell anemia to avoid dehydration, extreme temperature or physical exertion/exhaustion, high altitudes, and recreational drugs that may affect the heart. If someone is having a pain crisis, hydration and medications can be used to help manage the pain and risk for related complications.
There are also some screening tests that are recommended for people with sickle cell anemia that can include blood work and imaging such as Doppler ultrasound, chest x-ray, ECG, and abdominal ultrasound. Women who have sickle cell anemia that are pregnant can have an increased risk for preterm labor, blood clots, and infections, so it is important that they work closely with their medical team to make sure they taking all the necessary precautions.
A research study that was done in 1994 estimated that the average person with sickle cell anemia lives to be in their 40s. However, some more recent studies have shown that as our understanding and treatment of sickle cell anemia improves, there has been shift toward a longer lifespan and fewer childhood deaths. There are also more treatments that are being developed that will likely further increase the life expectancy of people who have sickle cell anemia.
Spinal Muscular Atrophy (SMA) is an inherited genetic condition that mainly affects muscle movement. It is characterized by progressive weakness and wasting of muscles due to a loss of motor neurons involved in muscle movement control. Motor neurons are needed to transmit signals from the brain and spinal cord to the muscles, which tells the muscles when to contract, which allows the body to move.
People with SMA have muscle weakness that typically worsens with increasing age. Muscles that are closer to the center of the body (proximal) are typically more affected than muscles that are further away from the center of the body (distal). In additional to muscle weakness and wasting of muscles, people with SMA can also experience poor weight gain and growth failure, restrictive lung disease, scoliosis, joint contractures, and sleep difficulties. SMA is not thought to impact the brain directly, so generally people with SMA have normal intelligence and cognitive functioning.
There are many types of SMA, broken down by the age of onset as well as the severity of the muscle weakness.
SMA Type 0: The rarest and most severe form of SMA that is evident before birth. Pregnancies that are affected with SMA type 0 generally appear to show less fetal movement, often resulting in joint deformities (contractures) at birth. Some infants may also have congenital (from birth) heart defects. Affected infants have severe muscle weakness and low muscle tone (hypotonia), especially the muscles in the respiratory system. Because of this, most infants with SMA type 0 do not survive past infancy due to respiratory failure.
SMA Type 1: The most common form of SMA, and babies with SMA type 1 can typically be identified at birth or shortly thereafter. Affected infants are unable to control head movements or sit independently. They may also experience swallowing difficulty. Weakness of the respiratory muscles often leads to respiratory failure. Most infants and children with SMA type 1 do not survive past early childhood.
SMA Type 2: Muscle weakness develops in children with SMA type 2 between 6 to 12 months. Affected children can typically sit unassisted but may required help getting into the seated position. Their weakness, however, is progressive and becomes worse later in childhood. They are unable to stand or walk unaided, and can often also have scoliosis, tremors of the fingers, and weakness of the respiratory muscles that can be fatal. The lifespan of these affected individuals varies but many are able to live into their twenties or thirties.
SMA Type 3: Develops after 12 months of age, with onset after affected children have started standing and walking independently. Muscle weakness predominantly affects the lower extremities before progressing to the upper limbs. Difficulty walking or climbing stairs is common, however some affected individuals retain the ability to walk into adulthood. Scoliosis and finger tremors are also common, with less respiratory involvement than types 1 or 2. Life expectancy is normal or near-normal.
SMA Type 4: This adult-onset form is typically diagnosed in the third decade of life, presenting with gradual muscle weakness affecting the legs and hips before progressing to the shoulders and arms. Mobility is often affected, with a ‘waddling gait’ being fairly common, and affected individuals may eventually require the use of a wheelchair or other assistance. Finger trembling and muscle twitches may also occur. Life expectancy is normal.
In the past, individuals were classified into these different subtypes based on their physical presentation. With our increasing understanding of the genetics behind SMA, we are finding that there may be an even wider spectrum of how severely someone with SMA is affected. Combining someone’s physical presentation with their genetics can sometimes make it difficult to classify a patient clearly into a sub-type.
Causes of Spinal Muscular Atrophy
We have over 20,000 different genes in the body. These genes are like instruction manuals for how to build a protein, and each protein has an important function that helps to keep our body working how it should. The SMN1 gene makes a protein called the survival motor neuron (SMN) protein. The SMN protein is found throughout the body, but is the most concentrated in the spinal cord, and it works to keep special nerve cells (called motor neurons) working. These motor neurons are responsible for carrying signals from the brain and the spinal cord to our muscles that tell them to contract, which allows the body to move.
If someone has a harmful change (called a pathogenic variant) in both of their SMN1 genes (the one they got from their mom and the one they got from their dad), then their body is not going to make enough of the SMN protein. If the body does not have enough SMN protein, then the motor neurons cannot transmit the signals from the brain and spinal cord to the muscles in the body as easily. Without signals to tell the muscles to contract, the muscles can become weaker and waste away. This is what leads to the symptoms we associate with SMA.
In addition to the SMN1 gene, the number of SMN2 genes that someone has can be helpful to try to predict the severity of the condition. Like the SMN1 gene, the SMN2 gene is responsible for making SMN proteins. However, the amount of SMN proteins made by the SMN2 gene is relatively small compared to the SMN1 gene. While we all have two copies of the SMN1 gene, the number of SMN2 genes that people have varies, ranging from one to eight copies. Having multiple copies of the SMN2 gene and the SMN proteins that they make can help to make up for the lack of SMN proteins caused by the non-working SMN1 genes. This is believed to be associated with less severe symptoms of SMA, but the exact relationship between the number of SMN2 genes and the severity of the SMA symptoms is not fully understood yet.
SMA is inherited in an autosomal recessive pattern. This means an individual who has SMA has inherited two non-working copies of the SMN1 gene; the one they inherited from their mom is not working and the one they inherited from their dad is not working. In the case of autosomal recessive conditions, if you inherit one working SMN1 gene from a parent and one non-working SMN1 gene from a parent, you are called a ‘carrier’ for SMA. Carriers do not have SMA, and typically do not have signs or features of SMA.
It is estimated that approximately 1 in 8000 to 1 in 10,000 people have spinal muscular atrophy. About half of these will have SMA type 1.
Treatment/Management for Spinal Muscular Atrophy
Management of SMA should include an assessment of the patient’s nutritional state, their respiratory function, and an evaluation of their bones. If nutrition is a concern, placement of a gastrostomy tube (G-tube) may be recommended. Consultation with a pulmonologist familiar with SMA is also recommended, especially if issues with respiratory function become a problem. Children with SMA should have an evaluation that looks at their muscles and bones to see if they are having any difficulties that may be improved with additional therapies or services, such as a wheelchair or orthotic inserts.
Research on SMA has focused on increasing the body’s production of SMN proteins, since the absence of this is what causes SMA. In 2017, the U.S. Food and Drug Administration (FDA) approved a treatment called antisense therapy for SMA, which basically changes someone’s SMN2 genes so that they can produce more of the SMN protein. Because this therapy is designed to treat SMA by going directly to the cause of it, this medication could potentially be effective at slowing or stopping the symptoms of SMA.
Conditions that may be diagnosed prenatally
The amount of genetic and chromosome conditions that can be tested for in pregnancy is constantly evolving. One group of conditions is Sex Chromosome Variations that result when a baby has a different number of sex chromosomes, also known as X & Y chromosomes, then we would expect to see in some or all of the cells in their body. A secondary condition group called Microdeletion Syndromes occurs when a small area of a chromosome (which is made up of our DNA) is deleted or missing.
Cri-du-chat syndrome (5p) is classified as a Microdeletion Syndrome. Each individual with Cri-du-chat syndrome is unique. Most individuals with Cri-du-chat syndrome have some degree of development and intellectual delay, and are usually moderately to severely affected. Other possible health concerns include congenital (born with) heart defects, seizures, scoliosis, and the development of a hernia in the lower abdomen (inguinal hernia).
People with Cri-du-chat syndrome can have loving relationships with friends and family, and learn and make progress in their social skills and communication at their own pace. Most individuals with Cri-du-chat syndrome will need support and care over their lifetime given the related medical issues.
Causes of Cri-du-chat Syndrome
Cri-du-chat syndrome is caused by a deletion on the short arm (p) of chromosome 5. The symptoms of Cri-du-chat syndrome are thought to be caused by the loss of multiple genes on this chromosome due to the deletion. Most of the time, this deletion is said to be ‘de novo‘, which means that it was not passed down from a parent; it is brand new in the individual who is diagnosed with the condition. In these cases, the deletion occurs in the development of an egg or a sperm and is then copied over into every cell in the developing baby’s body.
In some cases (10-12%), Cri-du-chat syndrome can be inherited when one parent’s chromosome 5 is part of a balanced translocation. A balanced translocation occurs when a piece of chromosome 5 breaks off and trades places with a piece of another chromosome. No genetic material is lost when this happens; the chromosome pieces just trade places. Because the parent has not lost any genetic material, they do not show symptoms for any genetic condition. However, when they have children, their egg or sperm can be missing genetic material from chromosome 5 leading to a higher chance of Cri-du-chat. The specific chance for Cri-du-chat depends upon the chromosome segments involved in the parent’s translocation.
Concerns Associated with Cri-du-chat syndrome
Cri-du-chat syndrome is associated with a variety of health and developmental concerns. Small head size (microcephaly), low birth weight, delayed growth, and low muscle tone (hypotonia) are some of the early symptoms of Cri-du-chat syndrome, as well as a high-pitched, cat-like cry. Delayed development, delayed or absent speech, and intellectual disability are also found in individuals with Cri-du-chat syndrome. Some babies may be born with a heart defect. Scoliosis and seizures are other common health concerns associated with Cri-du-chat syndrome. Many individuals with Cri-du-chat syndrome also develop behavioral issues, such as self-harming or aggressive behaviors.
If I have a baby with Cri-du-chat syndrome, what is the chance I will have another baby with this condition
Because most cases of Cri-du-chat syndrome are caused by a de novo deletion on chromosome 5, the chance of having another child with Cri-du-chat syndrome is expected to be small. However, if the de novo deletion occurred early enough to be present in more than one egg or sperm, there could be a slight increased risk to have another child with the condition.
If Cri-du-chat syndrome was inherited from a parent with a chromosome 5 translocation, the chance of having another child with Cri-du-chat depends upon the specific pieces of chromosomes exchanged. If you are found to have a chromosome translocation, meeting with a specialist, such as a genetic counselor, can help to better understand what your specific risks may be.
How do parents of children with Cri-du-chat syndrome feel about raising a child with it?
Many families of children with Cri-du-chat share valuable information and support with each other through the Five P Minus Society. Their web site has lots of helpful information for families with a member who has Cri-du-chat syndrome.
Treatment/Management for Cri-du-chat syndrome
There is no cure for Cri-du-chat syndrome. However, medical management and therapeutic intervention can help individuals reach their full potential. Individuals with Cri-du-chat syndrome will require regular follow-up and treatment by a variety of health specialists to treat their specific needs. Intervention programs such as physical therapy, speech therapy, occupational therapy, and behavioral therapy can be beneficial to individuals with Cri-du-chat syndrome.
What are the long-term outcomes for individuals with Cri-du-chat syndrome?
It is important to recognize that the health and developmental effects of Cri-du-chat syndrome vary from person to person. Speech and mobility issues can be improved with intervention programs. Individuals with Cri-du-chat syndrome may have moderate to severe intellectual disabilities, and may benefit from special education. Most individuals with Cri-du-chat syndrome will need support and care over their lifetime given their health and developmental concerns. Life expectancy is expected to be normal for most individuals with Cri-du-chat syndrome.
Each individual with 1p36 microdeletion syndrome (also known as monosomy 1p36) is unique. Most individuals experience minor changes in physical appearance and intellectual and developmental disabilities of varying degrees.
1p36 microdeletion syndrome is one of the most common chromosome deletions. It is estimated that 1 in every 5,000 to 10,000 individuals have a 1p36 microdeletion; however, it is likely that many individuals who are affected do not get diagnosed.
Causes of Deletion Syndrome
1p36 microdeletion syndrome is caused by a deletion (“missing piece”) on the short arm (“p”) of chromosome 1 (which is where the 1p comes from; the 36 is the precise location on the chromosome that is missing). The deletion results in the loss of several genes. The size of the deletion can vary between individuals with the condition, and it is thought that the larger sized deletions lead to more severe symptoms. Most of the time this deletion is said to be ‘de novo’, which means that it was not passed down from a parent; it is brand new to that person in the family. In these cases, the 1p36 microdeletion occurs in the development of an egg or a sperm. About 5-10% of people with 1p36 microdeletion syndrome have inherited the deletion from an unaffected parent with a balanced chromosome rearrangement (called a translocation) that has a break at the 1p36 region.
A balanced translocation occurs when a piece of chromosome 1 breaks off and trades places with a piece of another chromosome. No genetic material is usually lost when this happens; the chromosome pieces just trade places. Because the parent usually has not lost any genetic material, they do not show symptoms for any genetic condition. However, when they have children, their egg or sperm can be missing genetic material from chromosome 1 leading to a higher chance of 1p36 microdeletion syndrome.
Concerns Associated with Deletion Syndrome
1p36 microdeletion syndrome is associated with a variety of health and developmental concerns; however, individuals with the condition can share many of the following characteristics: low muscle tone (hypotonia), growth and feeding problems, small head size (microcephaly), a prominent forehead, deep-set eyes with straight eyebrows, low set ears, a small mouth, a long area between the nose and mouth (philtrum), and a small pointed chin, among other differences in physical features. Seizures occur in more than half of individuals and many can also have structural differences of the brain. People with this condition may also experience vision and/or hearing problems. Differences of the skeletal, gastrointestinal system, heart, kidneys, or genitalia can also be observed. Many individuals with 1p36 microdeletion syndrome also develop behavioral issues, such as self-harming or aggressive behaviors. Individuals are expected to need extra help and support with learning and development.
What is life like for people with 1p36 microdeletion syndrome?
People with 1p36 microdeletion syndrome can have loving relationships with friends and family, and learn and make progress in their social skills and communication at their own pace. Most individuals with 1p36 microdeletion syndrome will need support and care over their lifetime given the related medical issues.
If I have a baby with 1p36 microdeletion syndrome, what is the chance I will have another baby with this condition?
The chance to have another baby with 1p36 microdeletion depends on the chromosome status of the parents. If both parents are found to have the typical number of chromosomes without any type of rearrangement, the odds for having another baby with 1p36 microdeletion are assumed to be low (less than 1%). If one of the parents is found to have a balanced translocation involving the 1p36 region, the odds to have another affected pregnancy will be higher. These odds will depend on the specific type of translocation observed in the parent. Parents could consider meeting with a genetic counselor to discuss the specific chances and options for prenatal screening and testing or pre-implantation genetic testing.
How do parents of children with 1p36 microdeletion syndrome feel about raising a child with the condition?
Many families of children with 1p36 microdeletion syndrome share valuable information and support with each other through the 1p36 Deletion Support and Awareness. Their website has lots of helpful information for families with a member who has 1p36 microdeletion syndrome.
Treatment/Management for Deletion Syndrome
There is no cure for 1p36 microdeletion syndrome. However, medical management and therapeutic intervention can help individuals reach their full potential. Individuals with 1p36 microdeletion syndrome will require regular follow-up and treatment by a variety of health specialists to treat their specific needs. Intervention programs such as physical therapy, speech therapy, occupational therapy, and behavioral therapy can be beneficial to individuals with this condition.
What are the long term outcomes for individuals with 1p36 microdeletion syndrome?
It is important to recognize that the health and developmental effects of 1p36 microdeletion syndrome vary from person to person. Speech and other developmental issues common to 1p36 microdeletion syndrome can be improved with intervention programs. Individuals with 1p36 microdeletion syndrome may have moderate to severe intellectual disabilities, and may benefit from special education. Most individuals with this condition will need support and care over their lifetime given their health and developmental concerns. Generally, individuals with the condition do survive well into adult life.
Other Resources
Unique: 1p36 microdeletion syndrome
1p36 Deletion Support & Awareness
Click here to learn more about scheduling a genetic counseling appointment for pregnancy-related questions.
DiGeorge syndrome (22q11.2) is classified as a Microdeletion Syndrome. DiGeorge syndrome, also called 22q11.2 deletion syndrome, is associated with several health and developmental concerns. It is important to remember that each individual with DiGeorge syndrome is unique, and the symptoms can vary tremendously from person to person. Some people with DiGeorge syndrome do not have any major noticeable health effects, and in fact may never know they have it, while others may have a number of significant health and developmental concerns which in some cases can be quite serious.
DiGeorge syndrome also goes by a couple other names, including velocardiofacial syndrome, Shprintzen syndrome, and conotruncal anomaly face syndrome. There are also a couple other health conditions, called autosomal dominant Opitz G/BBB syndrome and Cayler cardiofacial syndrome, that are more common in people with DiGeorge syndrome.
What is life like for people with DiGeorge syndrome?
People with DiGeorge syndrome can have loving relationships with friends and family, go to school with their peers, and are interested in learning about a variety of topics. As adults, many individuals work in jobs they find meaningful, and are often able to live independently or semi-independently. Many people with DiGeorge syndrome will have children of their own. You can find stories about children and adults with DiGeorge syndrome at 22q.org.
Causes of DiGeorge syndrome
DiGeorge syndrome is caused by a deletion on the long arm (“q”) of the 22nd chromosome, which is also why it is called 22q11.2 deletion syndrome (11.2 is specific location where the deletion is). This deletion results in the loss of several genes. Most of the time this deletion is said to be ‘de novo’, which means that it was not passed down from a parent; it is brand new in the individual who is diagnosed with the condition. In these cases, the deletion occurs in the development of an egg or a sperm and is then copied over into every cell in the developing baby’s body. About 10% of people who are diagnosed with DiGeorge syndrome have inherited the deletion from a parent who also has it. In many of these cases, the parent did not know they had DiGeorge until their child was diagnosed.
Concerns Associated with DiGeorge syndrome
The health and developmental concerns associated with individuals with DiGeorge syndrome varies significantly from person to person, even within the same family. Below is a list of some of the more common concerns associated with DiGeorge syndrome. It is important to know that typically people with 22q11.2 deletion syndrome do not necessarily experience all of these health concerns.
- Congenital (present from birth) heart defects
- Abnormalities of the palate (roof of the mouth), such as cleft palate
- Developmental delays, such as walking or talking later than normal
- Learning difficulties
- Mild intellectual disability
- Immune deficiency/autoimmune disorders
- Hypocalcemia (not enough calcium in the body)
- Difficulties with feeding and swallowing
- Gastrointestinal anomalies
- Hearing loss
- Growth hormone deficiency
- Seizures
- Ophthalmologic (eye) abnormalities
- Autism or autistic spectrum disorder
- Psychiatric illness in adults
If I have a baby with DiGeorge syndrome, what is the chance I will have another baby with this condition?
The chance to have another baby with DiGeorge syndrome depends on whether or not one of the parents carries the deletion. If one of the parents of an individual with DiGeorge syndrome is found to carry the deletion, the chance for each pregnancy to be affected is 50%. If both parents undergo genetic testing for the deletion and neither are found to carry it, then the risk to for future pregnancies is low in most cases.
How do parents of children with DiGeorge syndrome feel about raising a child with it?
A study that surveyed the parents of 76 school-aged children with DiGeorge syndrome reported that the majority of parents felt that their child had brought joy and happiness to their lives. They said they enjoyed their child’s sense of humor, and felt that having their child increased their patience and tolerance. Many parents indicated challenges regarding parenting a child with DiGeorge syndrome related to their learning, communication, and medical difficulties. Many parents expressed some concern about the future for their child related to their child’s ability to live independently, find meaningful employment, and concerns related to future mental and physical health.
Treatment/Management for DiGeorge syndrome
Typically, the chromosome deletion that causes DiGeorge is copied over into every cell in the person’s body, and the missing genes and proteins cannot be replaced. There is no “cure” for DiGeorge syndrome. Screening for and treatment of various health concerns can be important to reduce complications associated with this condition. Individuals with DiGeorge syndrome may require regular follow-up and treatment from various health specialties depending on their specific needs. In many cases, early intervention programs for individuals with DiGeorge syndrome can help them reach their full potential. Intervention programs may include speech, physical, occupational, and/or educational therapy.
What are the long term outcomes for individuals with DiGeorge syndrome?
The health and developmental effects of DiGeorge syndrome vary tremendously, so the long term prognosis is very individual. Some people with DiGeorge syndrome have no intellectual delays at all, while others may have more significant developmental and intellectual delays and may need more help. Some individuals with DiGeorge syndrome will develop mental health concerns, others will not. Physical health issues related to the heart, immune system, and endocrine system be ongoing issues throughout life, or they could not be diagnosed until adulthood. The information available on life expectancy is limited, however some studies indicate that individuals with DiGeorge syndrome may often have a shorter than average expectancy.
How common is DiGeorge syndrome?
Most studies indicate that the DiGeorge syndrome occurs in about 1 in 4,000 births. However, many think it may be more common given that a high percentage of people with DiGeorge syndrome may never be diagnosed.
Down syndrome, also known as trisomy 21, is a genetic condition caused by an extra copy of the 21st chromosome. Each individual with Down syndrome is unique. Individuals with Down syndrome have variable health issues, including developmental delays and some degree of intellectual disability.
What is life like for people with Down syndrome?
Individuals with Down syndrome can have loving relationships with friends and family, and can learn and make progress in their social skills and communication at their own pace. All individuals with Down syndrome have some degree of intellectual delay, although it is quite variable. Some people with Down syndrome are able to live semi-independently lives, holding meaningful jobs and being active in their community. Others may have more significant delays, and can require continued care throughout their lifetime.
Causes of Down syndrome
Down syndrome, also known as trisomy 21, is caused by having an extra copy of the 21st chromosome. Chromosomes are structures that hold our genes, which is the information that tells our bodies how to grow and function. As humans, we typically have a total of 46 chromosomes, or 23 pairs, one set inherited from our mother through the egg and one from our father through the sperm.
Chromosome conditions are caused when a baby has extra or missing chromosomes. These conditions usually do not run in families. There is nothing you can do to cause or prevent a chromosome condition. For many of these conditions, the chance increases with the age of the mother, but there are babies born with chromosome conditions to mothers of all ages. Pregnancies where the baby has a chromosome condition have a higher risk for miscarriage. In rare cases, Down syndrome occurs due to a chromosome translocation (when two pieces of chromosomes break off and switch places), which can increase the risk to have another pregnancy with Down syndrome. A chromosome translocation related to Down syndrome may also increase the chance for family members to have a pregnancy with Down syndrome.
Concerns Associated with Down syndrome
The health concerns associated with Down syndrome vary from person to person but may include:
- Heart defects, present at birth in approximately half of all babies with Down syndrome
- Low muscle tone (hypotonia), which affects approximately 80% of people with Down syndrome
- Blockage in part of the intestine or functional problem with part of the intestine (gastrointestinal defect), which affects approximately 12% of people with Down syndrome
- Hypothyroidism
- Vision problems
- Hearing problems
How common is Down syndrome?
The incidence of Down syndrome is approximately 1/700 births. The chance of having a baby with Down syndrome due to trisomy 21 increases as the mother’s age increases. Most cases of Down syndrome are sporadic, meaning they do not run in families. There is nothing we know of that can be done to cause or prevent Down syndrome from occurring.
If I have a baby with Down syndrome syndrome, what is the chance I will have another baby with this condition?
In cases where Down syndrome is due to trisomy 21 and not a chromosome translocation, the chance to have another baby with Down syndrome is approximately 1%. If your age-related risk at the time of the birth of the baby is higher than 1%, then that age-related risk would be the chance for the baby to have Down syndrome. There may be a much higher chance for recurrence of Down syndrome in families when it is caused by a balanced translocation carried by one of the parents. Keep in mind, it is quite rare for Down syndrome to be caused by a translocation. A picture of the baby’s chromosomes (karyotype) can determine if the Down syndrome is caused by trisomy 21 or the type that can run in the family due to a translocation.
Treatment/Management for Down syndrome
Sometimes surgery is needed to treat heart defects or abnormalities of the digestive tract present at birth. Other health conditions are treated as needed, such as hearing and vision concerns, and thyroid disorders.
Early intervention programs can help all individuals with Down syndrome reach their full potential. Intervention programs may include speech, physical, occupational, and/or educational therapy.
Federal law requires that children with Down syndrome receive an appropriate education in the public school system, and they may receive support services within the regular classroom. Many individuals with Down syndrome finish high school, and to date there are over 250 colleges offering programs for individuals with intellectual disabilities.
What would having a baby with Down syndrome mean for my family?
Raising a child with special needs, including Down syndrome, can result in more cost and may require more time and patience. However, research has shown that having a child with Down syndrome can have a positive effect on the family.
Living Life with Down syndrome
Each individual is unique, however, this is a list of some things that are possible for individuals with Down syndrome:
- Participate in community sports and activities
- Can be in mainstream/’regular’ classes at school
- Also may have special education classes
- May complete high school
- Can be employed competitively
- Independent living
- Living in a group home
- Have friends
- Have intimate relationships
Resources:
Trisomy 18, also known as Edwards syndrome, is a genetic condition caused by an extra chromosome 18. Babies with trisomy 18 generally have many complex medical complications, including heart defects, growth restriction, a small abnormally shaped head, and clenched fingers with overlapping fingers.
Because of the severity of medical complications associated with trisomy 18, only about 50% of babies with trisomy 18 will survive to delivery. About 5-10% of babies with trisomy 18 will live past the first year of life, with a much smaller percentage surviving to adulthood. All of these individuals will have severe intellectual disability, and will have significant medical issues that will require full-time caregiving.
Finding out your pregnancy has trisomy 18 can be very difficult. Determining what to do next can be even more challenging. Some people who know that their pregnancy has trisomy 18 would choose to carry the pregnancy to term, and use the information to be prepared for the baby’s arrival. Others would choose to terminate (or abort) a pregnancy that has many significant medical complications.
There is no right or wrong answer; only the answer that is right for you and your family. GSF aims to provide you with the information you need to make that difficult decision, and are here to support you in any way that we can.
Find more information about trisomy 18 at the Trisomy 18 Foundation’s website.
Jacob's syndrome (47,XYY) is classified as a Sex Chromosome Variation. Each individual with Jacob’s syndrome is unique. Some individuals with Jacob’s syndrome may be above average height, but Jacob’s syndrome does not affect other physical features. Sexual development is also normal. Individuals with Jacob’s syndrome have an increased risk of learning disabilities, delayed speech, and behavioral problems. A small number of those with Jacob’s syndrome are also diagnosed with autism spectrum disorders. The symptoms of Jacob’s syndrome vary greatly from person to person, but they are generally mild and some individuals with Jacob’s syndrome may remain undiagnosed.
What is life like for people with Jacob’s syndrome?
People with Jacob’s syndrome can have loving relationships with friends and family, go to school with their peers, and develop their social skills and communication. Individuals with Jacob’s syndrome live healthy, full, and independent lives. Most men with Jacob’s syndrome are able to have children of their own.
Causes of Jacob’s syndrome
In a typical genome, individuals have two sex chromosomes to determine biological sex. Women usually have two X chromosomes (one from mom and one from dad), and men usually have one X chromosome (from mom) and one Y chromosome (from dad). Individuals with Jacob’s syndrome have one X chromosome and two Y chromosomes, so they have 47 chromosomes total and their sex chromosomes are XYY. Jacob’s syndrome is usually caused by a cell division error in the sperm before conception. Rarely, the cell division error may occur after conception. These individuals may have mosaic Jacob’s syndrome, meaning some of their cells are XYY and some are XY.
Concerns Associated with Jacob’s syndrome
Jacob’s syndrome is associated with few health or developmental concerns. Increased height is one of the main symptoms of Jacob’s syndrome and may be noticeable as early as 5 years old. Jacob’s syndrome does not cause infertility or abnormal sexual development. Speech delays and learning disabilities such as dyslexia occur in some individuals with Jacob’s syndrome. Some individuals with Jacob’s syndrome may also develop behavioral issues. There is a small increased risk of autism spectrum disorders associated with Jacob’s syndrome.
If I have a baby with Jacob’s syndrome, what is the chance I will have another baby with this condition?
Most cases of Jacob’s syndrome are not inherited, but are caused by a cell division error in the sperm before conception. Mosaic Jacob’s syndrome is also not inherited. The chance of having another baby with Jacob’s syndrome does not increase in these cases.
Treatment/Management for Jacob’s syndrome
There is no “cure” for Jacob’s syndrome, but therapeutic intervention can help individuals struggling with learning disabilities or behavioral issues reach their full potential. Speech therapy can be beneficial to individuals with speech delays. School support for learning disabilities and/or educational therapy can also be beneficial. Individuals with Jacob’s syndrome tend to respond well to intervention measures, and in some individuals these issues may resolve completely.
What are the long-term outcomes for individuals with Jacob’s syndrome?
It is important to recognize that the health and developmental effects of Jacob’s syndrome vary from person to person. Therapy and other interventions such as speech therapy and support for learning disabilities can be beneficial to individuals with Jacob’s syndrome. Men with Jacob’s syndrome are expected to live full, independent lives.
How common is Jacob’s syndrome?
Jacob’s syndrome is estimated to occur in 1 in 1,000 newborn boys.
Jacobsen syndrome (11q) is classified as a Microdeletion Syndrome. Each individual with Jacobsen syndrome is unique. All individuals with Jacobsen syndrome have some degree of developmental and intellectual disabilities. Other common health concerns include a blood clotting disorder called Paris-Trousseau syndrome and abnormalities in the heart and bones.
What is life like for people with Jacobsen syndrome?
People with Jacobsen syndrome have loving relationships with friends and family, and learn and make progress in their social skills and communication at their own pace. Individuals with Jacobsen syndrome are expected to need support and care over their lifetime given the associated health concerns.
Causes of Jacobsen syndrome
Jacobsen syndrome is caused by a deletion on the long arm (also called the “q” arm) of the 11th chromosome. This deletion results in the loss of several genes. Most of the time (85%) this deletion is said to be “de novo”, meaning that it was not passed down from a parent and it is brand new in the individual who is diagnosed with the condition. In this case, the deletion occurs in the development of an egg or a sperm and is then copied over into every cell in the developing baby’s body.
In some cases (5-15%), Jacobsen syndrome can be inherited when one parent’s chromosome 11 has been changed by a “balanced translocation.” A balanced translocation occurs when a piece of chromosome 11 trades places with a piece of another chromosome. No genetic material is deleted during this process, the chromosome pieces just trade places. Because the parent has not lost any genetic material, they would not have Jacobsen syndrome. This translocation can become unbalanced when it is passed on, leading to Jacobsen syndrome. In an unbalanced translocation, some genetic material is lost when the parental chromosomes are passed down to the baby. Individuals who have Jacobsen syndrome caused by an unbalanced translocation have lost some genetic material on the end of the long arm (q) of chromosome 11.
Concerns Associated with Jacobsen syndrome
Jacobsen syndrome is associated with a variety of health and developmental issues. Infants with Jacobsen syndrome often have feeding difficulties and delayed development. Most individuals with Jacobsen syndrome have some degree of intellectual impairment, and many individuals with Jacobsen syndrome are diagnosed with attention-deficit hyperactivity disorder (ADHD). Behavioral issues such as compulsive behavior are also common. Crossed-eyes and drooping eyelids cause vision problems in many individuals with Jacobsen syndrome, but these issues can be corrected by surgery. Many individuals with Jacobsen syndrome also have skeletal abnormalities like a curved spine (scoliosis). One of the most serious health concerns associated with Jacobsen syndrome is abnormal development of the heart. A portion (around 20%) of individuals with Jacobsen syndrome do not survive early life because of severe heart defects. Around 90% of individuals with Jacobsen syndrome also have a bleeding disorder called Paris-Trousseau syndrome, which causes easy bruising and bleeding.
If I have a baby with Jacobsen syndrome, what is the chance I will have another baby with this condition?
The chance of having another baby with Jacobsen syndrome depends on the type of genetic change that caused Jacobsen syndrome in the individual. In cases where Jacobsen syndrome is caused by a “de novo” deletion, there is not a significant chance of having another child with Jacobsen syndrome. When a parent has Jacobsen syndrome, there is a 50% chance of having a child with Jacobsen syndrome. If a parent has a balanced translocation on chromosome 11, there is a higher chance of having another child with Jacobsen syndrome.
Treatment/Management for Jacobsen syndrome
There is no “cure” for Jacobsen syndrome. However, medical management can help individuals reach their full potential. Individuals with Jacobsen syndrome will need regular follow-up and treatment by a variety of health specialists to treat their specific needs. Serious heart abnormalities may require surgical intervention, and surgery to correct skeletal abnormalities may improve quality of life. Surgery may also be performed to improve vision in individuals with Jacobsen syndrome when crossed eyes and drooping eyelids are impairing vision. Because many individuals with Jacobsen syndrome have a bleeding disorder called Paris-Trousseau syndrome, special care needs to be taken during surgery. Therapeutic interventions such as occupational, speech, physical, and behavioral therapies can be beneficial to individuals with Jacobsen syndrome.
What are the long-term outcomes for individuals with Jacobsen syndrome?
It is important to recognize that the health and developmental effects of Jacobsen syndrome vary from person to person. Around 20% of individuals with Jacobsen syndrome do not survive the first 2 years of life, usually because of severe heart abnormalities. Special medical attention is needed during early life to avoid complications from heart abnormalities or bleeding, but people with Jacobsen syndrome are expected to need care and support over their lifetime. Life expectancy for individuals who survive early life is unknown, but individuals with Jacobsen syndrome have lived to adulthood.
How common is Jacobsen syndrome?
Jacobsen syndrome is believed to affect 1 in 100,000 people.
How do parents of children with Jacobsen syndrome feel about raising a child with Jacobsen syndrome?
Parents’ experiences with Jacobsen syndrome are summarized in this document, "Having a Son or Daughter with Jacobsen Syndrome/11q Deletion Syndrome: Perspectives of Parents”
Additional Resources:
Lettercase.org - Part of the National Center for Prenatal an Postnatal Resources
Klinefelter (47,XXY) syndrome is classified as a Sex Chromosome Variation. 47,XXY occurs when a boy has an extra copy of an X chromosome in some or all of his cells. It usually does not cause them to look physically different than their peers, but does cause a variety of subtle physical differences including:
- taller than average height
- lower muscle tone
- impaired fertility
Other features that can be seen include a curved little finger, difficulty fully straightening the elbows, flat feet, and a small depression in the chest.
Individuals with 47,XXY typically have intelligence in the normal range, although some learning disabilities and speech/expressive language delay are common. Some individuals with Klinefelter syndrome have more significant health and developmental concerns, while others may have few, if any, notable features, and may not even know that they have the condition.
Causes Klinefelter syndrome
47,XXY occurs when either the sperm or egg that make a pregnancy has an extra sex chromosome, or when there are errors in cell division early in a pregnancys development. The exact reason for these occurrences is not known.
Concerns Associated with Klinefelter syndrome
During pregnancy, the condition may be diagnosed through genetic testing such as a chorionic villus sampling (CVS) or amniocentesis, or suspected through a blood test such as cell-free DNA (cfDNA) analysis. Keep in mind that cfDNA cannot tell for certain if a baby has a variation in their number of X and Y chromosomes. It is important to remember that the signs and symptoms of Klinefelter syndrome can vary quite a bit from person to person. While prenatal diagnostic testing can provide a ‘yes’ or ‘no’ answer about whether a pregnancy has Klinefelter syndrome, it can not predict the specific health concerns that a baby with Klinefelter might have due to the variability of the condition.
During the newborn period, boys with Klinefelter syndrome are often no different from other boys. During early childhood, low muscle tone and/or a delay in meeting developmental milestones may or may not be seen. Between the ages of 5 and 8 they may often have an increase in height above their peers, and may also have longer arms and legs. During school years, there may also be a lag in language skills and academic difficulties.
Many boys with Klinefelter syndrome appear to enter puberty normally but have a tendency for testosterone levels to decline in late adolescence and early adulthood. Therefore, testosterone supplements may be given to fully develop secondary sexual characteristics, such as muscle patterns, a deeper voice, and facial and body hair.
Most men with Klinefelter syndrome have infertility. However, there have been reports of men fathering natural pregnancies and pregnancies with assisted medical technologies. Most of these cases have occurred in men that have a mixture of 47,XXY and 46,XY cells (mosaicism).
As men with Klinefelter syndrome grow older, there is some higher risk for a range of conditions including osteoporosis, thyroid disorders, diabetes, and autoimmune diseases. They also have an increased risk of acquiring leg ulcers.
If I have a baby with Klinefelter syndrome, what is the chance I will have another baby with this condition?
In most cases, the chance that a woman will have another baby with an X and Y chromosome variation is thought to be less than 1%, as the majority of cases of X and Y chromosome variation are not inherited.
Treatment/Management for Klinefelter syndrome
There is no “cure” for Klinefelter syndrome. Babies with Klinefelter syndrome often do not need any special treatment or intervention as infants. If any symptoms, such as learning disabilities, arise during childhood, they can be treated and managed with early intervention and support in school.
Many teens and adults with 47,XXY are given testosterone supplements to support full development of male secondary sex characteristics. Testosterone also helps increase and maintain bone density, but is unable to reverse infertility. Therefore, children who are reaching adolescence should work with a pediatric endocrinologist to determine if hormone therapy is recommended.
How common is Klinefelter syndrome?
47,XXY (Klinefelter syndrome) is the most common variation in X and Y chromosomes and is estimated to occur in one of every 500 males.
Living Life with Klinefelter syndrome
Most men with Klinefelter syndrome will be taller than average and have infertility. Some may experience difficulty in learning in some areas, however many individuals with Klinefelter syndrome have success in school, including higher education. While many have issues with infertility some are able to have children with or without reproductive assistance, while others will go on to adopt children. Individuals with Klinefelter syndrome may need monitoring of their health throughout their life, but can expect to live full and productive lives.
Additional Resources:
lettercase.org - Part of the National Center for Prenatal and Postnatal Resources
Langer-Giedion syndrome (8q) is classified as a Microdeletion Syndrome. Each individual with Langer-Giedion syndrome is unique. Most individuals with Langer-Giedion syndrome have some degree of developmental and intellectual delay. Other common health concerns include the development of bone tumors (osteochondromas) that can cause pain, limit the range of motion in joints, and put pressure on blood vessels and nerves.
What is life like for people with Langer-Giedion syndrome?
People with Langer-Giedion syndrome can have loving relationships with friends and family, and learn and make progress in their social skills and communication at their own pace. Some individuals with Langer-Giedion syndrome are able to live semi-independently, while those with more severe symptoms may need support and care over their lifetime.
Causes Langer-Giedion syndrome
Langer-Giedion syndrome is caused by a deletion or genetic change on chromosome 8. Two missing genes, EXT1 and TRPS1, cause some of the symptoms seen in Langer-Giedion syndrome. There may be additional deleted genes causing Langer-Giedion syndrome that have not been identified yet. Most of the time, this deletion is said to be ‘de novo’, which means that it was not passed down from a parent; it is brand new in the individual who is diagnosed with the condition. In this case, the deletion occurs in the development of an egg or a sperm and is then copied over into every cell in the developing baby’s body.
Concerns Associated with Langer-Giedion syndrome
Langer-Giedion syndrome is associated with a variety of health and developmental concerns, including frequent upper respiratory infections such as sinus infections, sore throats, and tonsil infections. Individuals with Langer-Giedion syndrome tend to be shorter than average, and may have a variety of other skeletal and limb differences. These differences include joined fingers (syndactyly), cone-shaped ends of their long bones, and the development of bone tumors (osteochondromas). These bone tumors are noncancerous, but can cause pain and put pressure on blood vessels, nerves, and the spinal cord. These bone tumors can also limit range of motion if they develop in joints. Some individuals with Langer-Giedion syndrome also develop Coxa plana, which is associated with decreased blood flow at the top of the femur (the large bone in your thigh) that causes this part of the bone to break down. This can cause pain when walking and limit range of motion in the hip joint. The bone can regrow, but the breakdown may happen repeatedly. Low muscle tone (hypotonia), a wandering eye, delayed speech, and mild to moderate intellectual delay are common in individuals with Langer-Giedion syndrome.
If I have a baby with Langer-Giedion syndrome, what is the chance I will have another baby with this condition?
Because most cases of Langer-Giedion syndrome are caused by a de novo deletion on chromosome 8, it is expected that the chance to have another child with Langer-Giedion syndrome is small. However, if the de novo deletion occurred early enough to be present in more than one egg or sperm, there could be a slight increased risk to have another child with the condition.
If Langer-Giedion syndrome was inherited from a parent with a chromosome 8 deletion, there is a 50% chance of having another child with Langer-Giedion syndrome.
Treatment/Management for Langer-Giedion syndrome
There is no cure for Langer-Giedion syndrome. However, medical management and therapeutic intervention can help individuals reach their full potential. Individuals with Langer-Giedion syndrome will need regular follow-up and treatment by a variety of health specialists to treat their specific needs, and surgery may be required to remove bone tumors. Intervention programs such as physical therapy and speech therapy can also be very beneficial to individuals with Langer-Giedion syndrome.
What are the long-term outcomes for individuals with Langer-Giedion syndrome?
It is important to recognize that the health and developmental effects of Langer-Giedion syndrome vary from person to person. Speech and mobility issues can be improved with intervention programs. Individuals with Cri-du-chat syndrome may have moderate to severe intellectual disabilities, and may benefit from special education. Some individuals with Langer-Giedion syndrome are able to live semi-independently, while others may require more care. Life expectancy is expected to be normal for most individuals with Langer-Giedion syndrome.
How common is Langer-Giedion syndrome?
Langer-Giedion syndrome is very rare; there are no estimates of how often this syndrome occurs.
Open neural tube defects (ONTDs), which includes spina bifida and anencephaly, is a range of birth defects that can include the brain and/or spinal cord. This is generally something that happens very early in pregnancy, and can vary widely in severity and expected outcome. Although the cause of most cases of ONTDs are not known, there are a few known risks factors, such as maternal obesity, uncontrolled maternal diabetes, and exposure to certain antiseizure medications. Taking folic acid (a type of vitamin B) before and during pregnancy has been shown to reduce the risk for ONTDs during pregnancy.
ONTDs can often be diagnosed by ultrasound, but may also be identified by doing a diagnostic procedure, such as amniocentesis.
The two most common types of ONTDs are spina bifida and anencephaly. Spina bifida occurs when the bones in the spine do not fuse correctly. This can cause part of the spinal cord to bulge through the spine. There is a wide variability with spina bifida: some individuals may have a very small hole and may not even know they have it, while others can have more severe openings that cause significant medical complications. The size of the opening and where it is on the spine can sometimes give more information as to what to expect. To learn more about spina bifida, visit the Spina Bifida Association website.
While spina bifida can be quite variable, anencephaly is almost always life-limiting. Anencephaly is when all or part of the brain is missing. Most babies with anencephaly do survive to delivery, and those who do often pass away shortly after birth.
Trisomy 13, also known as Patau syndrome, is a genetic condition caused by an extra chromosome 13. Babies with trisomy 13 generally have many complex medical complications, including heart defects, brain and spinal cord abnormalities, very small or poorly developed eyes, cleft lip and/or cleft palate, and low muscle tone (hypotonia).
Because of the severity of medical complications associated with trisomy 13, more than 80% of babies do not survive past the first month of life. Children with trisomy 13 that survive infancy have severe developmental and intellectual disabilities, and may have an increased risk for certain types of cancer.
Finding out your pregnancy has trisomy 13 can be very difficult. Determining what to do next can be even more challenging. Some people who know that their pregnancy has trisomy 13 would choose to carry the pregnancy to term, and use the information to be prepared for the baby’s arrival. Others would choose to terminate a pregnancy that has many significant medical complications.
There is no right or wrong answer; only the answer that is right for you and your family. GSF aims to provide you with the information you need to make that difficult decision, and are here to support you in any way that we can.
Find more information about trisomy 13 at the Support Organization for Trisomies 18, 13, and related disorders (SOFT) on their website.
Trisomy X syndrome (47,XXX) is classified as a Sex Chromosome Variation. 47,XXX occurs when girls have an extra copy of an X chromosome in some or all of her cells. It does not cause them to look much different than their peers but does cause subtle physical differences including taller than average height, a curved little finger, a skin fold near the inner part of the eye and flat feet. Seizures, renal, genitourinary and cardiac abnormalities, and infertility can also be seen.
Individuals with 47,XXX typically have intelligence in the normal range, although some specific learning disabilities and speech/expressive language delay are common and can be supported. Some individuals with Trisomy X syndrome have more significant health and developmental concerns, while others may have few if any notable features and do not even know that they have the condition.
Causes of Trisomy X
47,XXX occurs when there are problems with formation of a parent’s egg or sperm or when there are errors in cell division after an egg and sperm meet to form an embryo. The exact reason for these occurrences is not known. The risk of trisomy X increases with advancing maternal age.
Concerns Associated with Trisomy X
It is important to remember that the signs and symptoms of 47,XXX can vary quite a bit from person to person. During pregnancy, the condition may be diagnosed through genetic testing such as a chorionic villus sampling (CVS) or amniocentesis or suspected through a blood test such as cell free DNA analysis. Keep in mind that cell free DNA analysis cannot tell for certain if a baby has a variation in their number of X and Y chromosomes.
During the newborn period, girls with 47,XXX are often indistinguishable from other girls. During early childhood, low muscle tone and/or a delay in meeting developmental milestones may or may not be seen. During school years, a lag in language skills and academic difficulties may or may not be seen.
Intelligence is typically in the normal or low-normal range but mild intellectual disability can be seen. Psychological features including attention deficits, anxiety and mood disorders are not uncommon. Occasional medical issues can include genito-urinary or kidney malformations, seizures and infertility. Autoimmune problems such as thyroid disorders may be more common in women with Trisomy X.
It is believed that the majority of women with 47,XXX have gone their entire lifetimes without ever knowing they have an extra X chromosome. However, more people are learning that they or their baby have the condition due to prenatal genetic testing or during infertility testing.
If I have a baby with 47,XXX (Trisomy X), what is the chance I will have another baby with this condition?
In most cases, the chance that a woman will have another baby with an X and Y chromosome variation is thought to be less than 1% as the majority of cases of X and Y chromosome variation are not inherited.
Treatment/Management for Trisomy X
There is no “cure” for 47,XXX as the genetic changes that cause the condition are typically present in some or all of the cells in the person’s body.
Individuals with 47,XXX often do not need any special treatment or intervention as infants. However, infants and children known to have triple X should have medical and developmental evaluations tailored to the features that can be associated with the condition for early detection if present. These include a renal ultrasound and cardiac evaluation as well as watch for staring spells or atypical movements. If any symptoms of 47,XXX such as learning disabilities or social/emotional difficulties arise during childhood, they should be managed with early intervention and support in school.
Teens and adult women with 47,XXX who experience menstrual irregularities or infertility should be evaluated for hormonal abnormalities that could signal ovarian insufficiency. Other autoimmune problems including thyroid problems should also be considered.
How common is Trisomy X?
47,XXX (Trisomy X) is the most common variation is estimated to occur in one of every 1000 females.
Living Life with Trisomy X
Most girls and women with 47,XXX will not be observably different from other girls and women. Some may be taller than average and some may have infertility. Some may experience difficulty in learning in some areas however, many individuals with 47,XXX have success in school, including higher education. Women with 47,XXX work in a variety of career settings. The majority of women with 47,XXX may have children on their own. Some may have children with the help of assisted reproductive technologies. Individuals with 47,XXX can expect to live full and productive lives.
Turner syndrome (45,X) is classified as a Sex Chromosome Variation. Turner syndrome (sometimes called 45,X or monosomy X) occurs when a girl has complete or partial absence of her second X chromosome in some or all of her cells. This missing chromosome causes them to be shorter than average in height and can affect how their ovaries work. Each girl and woman with Turner syndrome are unique, but are more likely than average to have other health issues including heart and kidney problems, autoimmune disorders, hearing and vision loss, and other health issues.
Individuals with Turner syndrome typically have normal intelligence, although some may have specific learning disabilities and speech or expressive language delays. Turner syndrome is variable, meaning some individuals have more significant health and developmental concerns while others have less.
Causes of Turner syndrome
The majority of Turner syndrome occurs when there are differences with formation of a parent’s egg or sperm. Some instances of Turner syndrome can be caused by errors that happen when the cells are dividing very early in a baby’s development. The exact reason for these occurrences is not known. While many girls and women with Turner syndrome will be missing the second X chromosome in all of the cells in their body, some will only be missing the second X chromosome in some of their cells. Girls and women with this mixture of cells have mosaic Turner syndrome, and may have fewer health or developmental issues than those who have non-mosaic Turner syndrome.
Less commonly, Turner syndrome occurs when there is loss of only a portion of a second X chromosome that can occur from a variety of mechanisms.
Concerns Associated with Turner syndrome
During pregnancy, it may be diagnosed through genetic testing such as a chorionic villus sampling (CVS) or amniocentesis or suspected through a blood test such as cell-free DNA (cfDNA) analysis. Keep in mind that cfDNA analysis cannot tell for certain if a baby has a variation in their number of X and Y chromosomes. It is important to remember that the signs and symptoms of Turner syndrome can vary quite a bit from person to person. While prenatal diagnostic testing can provide a ‘yes’ or ‘no’ answer about whether a pregnancy has Turner syndrome, it can not predict the specific health concerns that a baby with Turner syndrome might have due to the variability of the condition.
While many pregnancies with Turner syndrome will not appear different in any way, while some will show differences on prenatal ultrasound. These signs can include large collections of fluid at the back of a baby’s neck (an increased nuchal translucency or a cystic hygroma) or around the baby’s body (hydrops) as well as heart defects. For reasons that are unclear, the majority of pregnancies with Turner syndrome result in miscarriage. The miscarriage can occur in any of the three trimesters, but most often occurs in the first trimester.
Despite the high rate of miscarriage, the outlook for girls with Turner syndrome at birth is quite good. During the newborn period, some girls with Turner syndrome will no obvious signs or health issues, while others may show puffy hands and feet (edema), a short thick neck (webbed neck), and/or heart problems. Short stature is the most common physical difference for girls with Turner syndrome, and is usually apparent by school age. Early loss of ovarian function is also very common, causing many individuals with Turner syndrome to need hormone treatments to transition through puberty. Many women with Turner syndrome experience infertility as adults, but a small number do retain their ovarian function a bit longer and can conceive a child during young adulthood.
Other differences in development that can commonly be seen in girls and women with Turner syndrome include a low hairline at the base of the neck, skeletal differences, kidney problems, and hearing and vision loss. Autoimmune issues such as thyroid disorders and diabetes may also be more common in women with Turner syndrome.
Intelligence is typically in the normal range but nonverbal learning disabilities, developmental delays, and differences in behavior can be a little more common in girls with Turner syndrome.
If I have a baby with Turner syndrome, what is the chance I will have another baby with Turner syndrome?
In most cases, the chance that a woman will have another baby with an X and Y chromosome variation is thought to be less than 1%, as the majority of cases of X and Y chromosome variation are not inherited. However, some rare cases of Turner syndrome that involve the loss of a portion of a second X chromosome can potentially be passed from one generation to the next.
Treatment/Management for Turner syndrome
There is no “cure” for Turner syndrome. Many babies with Turner syndrome do not need any special treatment or intervention as infants. However, infants and children known to have Turner syndrome should have medical and developmental evaluations that look specifically for health issues that are associated with Turner syndrome, which can allow for early detection and changes to the baby’s medical management. These evaluations should include a heart evaluation and an ultrasound of the kidneys. Congenital (from birth) heart defects are identified in half of girls with Turner syndrome, and can be life-threatening.
School age girls with Turner syndrome should be seen by an endocrinologist (who specializes in hormones) to determine the optimal timing of hormone therapy to assist them through puberty and to reach a more typical final adult height. Regular hearing and vision screens as well as screens for diabetes, thyroid problems, and celiac disease are other common evaluations for girls with Turner syndrome. If any symptoms of learning disabilities or social/emotional difficulties arise during childhood, early intervention and support in school can be helpful.
Throughout adulthood individuals with Turner syndrome continue to receive hormone therapy, and should work with their providers to manage their overall health. This should include particular attention to blood pressure, heart, bone, and liver function.
Women with Turner syndrome have a decreased likelihood to conceive a pregnancy naturally. This chance also decreases quickly with age. Therefore, women with Turner syndrome who desire a family may not want to delay their family planning, or may wish to consider oocyte cryopreservation (freezing of their eggs) when possible. They may also wish to consider other options for motherhood such as egg donation, adoption, and gestational carriers (surrogates). Women with Turner syndrome that do carry a pregnancy have an increased risk for cardiovascular complications during the pregnancy.
How common is Turner syndrome?
Turner syndrome (45,X) is estimated to occur in one of every 2500 female births. It is unknown exactly how many pregnancies that result in miscarriage are affected with Turner syndrome.
Living Life with Turner syndrome
Many girls and women with Turner syndrome will be shorter than average in height and most will experience infertility. About half will also have heart problems that can in some cases be life-threatening. Some may experience difficulty in learning in some areas. However, many individuals with Turner syndrome have success in school, including higher education, and work in a variety of career settings. The majority of women with Turner syndrome will need coordinated medical care throughout their life, but can expect to live full and productive lives.
Resources
Lettercase.org: Part of the National Center for Prenatal and Postnatal Resources
Wolf-Hirschhorn syndrome (4p) is classified as a Microdeletion Syndrome. Each individual with Wolf-Hirschhorn syndrome is unique. Most individuals with Wolf-Hirschhorn syndrome have some degree of development and intellectual delay, and are anywhere from mildly to severely affected. Other possible health concerns include hearing loss, abnormalities in the heart, eyes, brain, skeleton, and urinary system, and seizures (which generally go away with age).
What is life like for people with Wolf-Hirschhorn syndrome?
People with Wolf-Hirschhorn syndrome can have loving relationships with friends and family, and learn and make progress in their social skills and communication at their own pace. Most individuals with Wolf-Hirschhorn syndrome will need support and care over their lifetime given the related medical issues.
Causes of Wolf-Hirschhorn syndrome
Wolf-Hirschhorn syndrome is caused by a deletion on the short arm (p) of chromosome 4. The symptoms of Wolf-Hirschhorn syndrome are believed to be caused by the loss of multiple genes on this chromosome due to the deletion. Most of the time (85-90%), this deletion is said to be ‘de novo’, which means that it was not passed down from a parent; it is brand new in the individual who is diagnosed with the condition. In this case, the deletion occurs in the development of an egg or a sperm and is then copied over into every cell in the developing baby’s body.
In some cases, Wolf-Hirschhorn syndrome can result from a genetic change causing chromosome 4 to become a ring chromosome. A ring chromosome is created when a chromosome breaks at both ends and the two ends fuse to form a circle. The loss of genetic material when this happens can cause Wolf-Hirschhorn syndrome.
In other cases, Wolf-Hirschhorn syndrome can be inherited when one parent’s chromosome 4 is part of a balanced translocation. A balanced translocation is when a piece of chromosome 4 breaks off and trades places with a piece of another chromosome. No genetic material is lost when this happens, the chromosome pieces just trade places. Because the parent has not lost any genetic material, they do not show symptoms for any genetic condition. However, when they have children, their egg or sperm can be missing genetic material from chromosome 4 leading to a higher chance of Wolf-Hirschhorn.
Concerns Associated with Wolf-Hirschhorn syndrome
Wolf-Hirschhorn syndrome is associated with a variety of health and developmental concerns, including:
- small head size (microcephaly)
- low birth weight
- delayed growth
- low muscle tone (hypotonia)
- distinctive facial features (called a “Greek warrior helmet”)
- most tend to be shorter than average due to delayed growth
- very delayed development of motor skills like sitting, standing, and walking
- delayed or absent speech
- mild to severe intellectual disability
- scoliosis
- hearing loss
- seizures
- some may be born with abnormalities in the heart, eyes, brain, skeleton, and urinary system
The severity of symptoms may be related to the amount of genetic material missing from chromosome 4.
If I have a baby with Wolf-Hirschhorn syndrome, what is the chance I will have another baby with this condition?
Because most cases (85-90%) of Wolf-Hirschhorn syndrome are caused by a de novo deletion on chromosome 4, there is not expected to be a high chance of having another child with it. However, if the deletion occurred early enough to be present in more than one egg or sperm, there could be a slight increased risk to have another child with the condition.
If Wolf-Hirschhorn syndrome was inherited from a parent with a chromosome 4 translocation, the chance of having another child with it depends upon the specific pieces of chromosomes exchanged. If you are found to have a chromosome translocation, meeting with a specialist, such as a genetic counselor, can help to better understand what your specific risks may be.
How do parents of children with Wolf-Hirschhorn syndrome feel about raising a child with it?
Many families of children with Wolf-Hirschhorn share valuable information and support with each other through the wolfhirschhorn.org. Their website has lots of helpful information for families with a member who has Wolf-Hirschhorn syndrome.
Treatment/Management for Wolf-Hirschhorn syndrome
There is no cure for Wolf-Hirschhorn syndrome. However, medical management and therapeutic intervention can help individuals reach their full potential. Individuals with Wolf-Hirschhorn syndrome will require regular follow-up and treatment by a variety of health specialists to treat their specific needs. Intervention programs such as physical therapy, special education, speech therapy, and occupational therapy can be beneficial to individuals with Wolf-Hirschhorn syndrome. Basic health care recommendations are available on 4p-supportgroup.org.
What are the long-term outcomes for individuals with Wolf-Hirschhorn syndrome?
It is important to recognize that the health and developmental effects of Wolf-Hirschhorn syndrome vary from person to person. Speech and mobility issues can be improved with intervention programs. Individuals with Wolf-Hirschhorn syndrome may have moderate to severe intellectual disabilities, and may benefit from special education. Medicine can also be used to manage seizures. Most individuals with Wolf-Hirschhorn syndrome will need support and care over their lifetime given their health and developmental concerns. Life expectancy is unknown for individuals with Wolf-Hirschhorn syndrome, but some individuals have lived into their 40’s.
How common is Wolf-Hirschhorn syndrome?
Wolf-Hirschhorn syndrome is believed to affect between 1 in 20,000 and 1 in 50,000 people.
Common Developmental Evaluations
When a baby or child begins showing signs of differences from their family background or their peers, their primary care provider may refer them for a developmental assessment. A genetics evaluation involves review of the child’s medical and family history, and detailed physical examination. Based on the review, genetic testing may be recommended. The results that come from genetic testing may provide your family with helpful information.
Autism spectrum disorder (ASD) is a complex developmental condition that can cause difficulties with communication, social interaction, and behaviors. Autism is referred to as a “spectrum” because of the variability in the effects and severity of symptoms that impact a person. The diagnosis of ASD is often made in early childhood, and is a lifelong condition. According to the Centers for Disease Control (CDC), about 1 in 59 children (1-2%) are affected by ASD.
Of note, in 2013, updates to the classification and diagnostic criteria for ASD were released (Diagnostic and Statistical Manual of Mental Disorders (DSM–5)). Those previously diagnosed with autistic disorder, Asperger’s syndrome, or pervasive developmental disorder not otherwise specified (PDD-NOS), are now all referred to as “autism spectrum disorder” or ASD.
Causes of Autism
In general, we do not fully understand the underlying cause for ASD. For the majority, it is likely caused by a combination of multiple genetic and environmental (nongenetic) factors (called multifactorial). Researchers have shown that about 20-40% of those with ASD will have an underlying genetic disorder, which means that they have a specific change in their genetic material that has contributed to their ASD. The remaining cases are likely due to a combination of factors. Examples of nongenetic risk factors include advanced parental age (when a man is 40 years old or older at the time of conception), short periods between pregnancies (less than 12-24 months), and pregnancy or delivery complications. In addition, the chances a child will have ASD increases if they have a family member with ASD.
For those who have an underlying genetic condition as the cause of their ASD, they may also have a chromosome abnormality or single gene disorder which can involve other health issues (such as PTEN Hamartoma Tumor syndrome). The likelihood of an underlying genetic condition may be impacted by whether a child has other features or health problems, such as distinctive physical features, birth defects, seizures, etc. This is often referred to as “syndromic” or “complex” ASD, and is more likely to have a genetic cause.
Genetic Testing for Autism
There are multiple types of tests that may be recommended for those who have ASD. The most common include chromosome microarray and fragile X testing. The chromosome microarray is used to identify small missing or extra pieces of chromosome material, and can identify or rule out several hundred known genetic syndromes. This testing has a detection rate (the chance for the testing to come back positive) of about 5-15% in those with ASD. Fragile X is a specific condition that can cause ASD, and accounts for less than 5% of those with ASD.
Further genetic testing that may be considered includes single gene testing, multigene panels, or whole exome sequencing. About 5-15% of individuals with ASD are found to have a single gene disorder that can be found with these types of tests.
Other testing that may be recommended includes biochemical screening, which involves measuring the levels of different substances in the blood or urine. These tests are used to look for metabolic conditions that can also cause ASD. These type of conditions account for less than 1% of those with ASD.
What is the chance to have another child with autism?
The chance to have another child with ASD (called the recurrence risk) can be dependent on whether an underlying genetic cause was identified. For instance, if the ASD in the family is determined to be caused by a genetic condition, the recurrence risk could be as high as 50%. In general, for a full sibling of an individual with ASD (regardless of other health conditions or if an underlying genetic cause was identified) the chance ranges from 7% to 20%. For families where there are two children with ASD the recurrence risk for a full sibling to be affected is estimated to be 25-35%. For other family members, such as half siblings, the chance is increased over the general population chance of 1-2%. It is also important to note that siblings of those with ASD are more likely to have language delays or autistic-like speech patterns, but may not have ASD.
Structural abnormalities, which are also called birth defects, are relatively rare and incredibly variable. They can range from very minor and easily treated to severe and even life-threatening. Some examples of birth defects are:
- Heart defects
- Cleft lip and/or palate
- Club foot
- Spina bifida
- Polydactyly (extra fingers or extra toes)
A birth defect can be an isolated finding, meaning that there are no other related health problems, or they could be an indicator of an underlying genetic condition or syndrome that may be associated with other health and developmental concerns.
What if a birth defect is detected on prenatal ultrasound?
The next steps will depend on the specific finding or concern. Sometimes a birth defect on ultrasound is associated with a higher likelihood of certain genetic or chromosomal condition in the baby and further diagnostic testing, such as amniocentesis, may be offered. While amniocentesis can provide a lot of information, there is a risk of miscarriage, so the decision about whether or not to undergo an amniocentesis is yours.
Regardless of whether or not you decide to do follow-up diagnostic testing and regardless of the results, it is not uncommon for more follow-up ultrasounds to take place after an ultrasound finding has been identified. It may be recommended that you deliver your baby at a hospital with a critical care nursery that is equipped to address any special medical needs your baby may have. Some birth defects can be repaired with surgery and some cannot.
In the rare situation that a baby is diagnosed with life threatening abnormalities on ultrasound, expectant parents should be counseled about their options. Some women choose to end their pregnancy if serious abnormalities are discovered. Some women decide to carry their pregnancies and may be supported through a perinatal hospice program if their baby is not expected to survive.
What if a birth defect is found after delivery?
Not all birth defects will be seen on prenatal ultrasound. Sometimes we don’t identify the specific finding or concern until after a baby is born. If a baby has a birth defect, the next steps will depend on the specific finding or concern. It may be recommended that your child be seen by a geneticist for further evaluation or undergo genetic testing (see Genetics & Child Development for more information).
Congenital hearing loss (or hearing loss from birth) is when someone has a reduced ability to hear sounds like other people do, and can range from mild to moderate to severe. Hearing loss is one of the most common birth defects in newborns, affecting approximately 1 to 2 of every 1000 babies every year. People who have hearing loss at birth or before the development of speech are said to have “prelingual” hearing loss. It is important to note that not all prelingual hearing loss is congenital. As we age the number of people diagnosed with hearing loss (called prevalence) increases with about 65% of those older than 70 being affected. Those who develop hearing loss after the development of normal speech have “postlingual” hearing loss.
There are three different types of hearing loss:
- Conductive hearing loss occurs when the outer or middle ear is affected and sound is not able to be transmitted through the ear. This may be due to a malformation or obstruction (e.g. fluid in the ears).
- Sensorineural hearing loss (SNHL) occurs when the inner ear is affected, and may be due to differences with the hair cells (sensory hearing loss), central auditory pathway (central loss), or when there is an abnormality in how sound is transmitted from the inner ear to the brain (auditory neuropathy spectrum disorder).
- Mixed hearing loss occurs when there is both conductive and sensorineural hearing loss.
Causes of Hearing Loss
Hearing loss can be caused by both hereditary (genetic) and environmental (nongenetic) factors, or due to a combination.
For those with congenital hearing loss about 50-60% will have an underlying genetic cause, which means that they have a specific change in their genetic material that has caused the hearing loss. In developed countries, this increases to about 80% for those with congenital hearing loss. The chances that hearing loss is genetic is impacted by age of onset, family history, laterality (one or both ears), type of hearing loss, and whether there are other features or health problems.
Common environmental factors that cause hearing loss include exposure to specific infectious agents either during the pregnancy (prenatal) or after (postnatal), exposure to certain medications (called ototoxicity), chronic ear infections or fluid in the ears (chronic otitis media), being born premature, significant noise exposure through occupation or recreation, injury to the ear, or aging. The most common infectious agents that cause congenital hearing loss include viruses called toxoplasmosis, rubella, cytomegalovirus (CMV), and herpes — all together these are called the TORCH “organisms.” Of these, CMV is the most common, and affected individuals may present either at birth or later in infancy with hearing loss.
Causes of Congenital Hearing Loss
Genetic causes of hearing loss may be classified as syndromic or non-syndromic. Syndromic means that the genetic cause is associated with a combination of other health problems or physical differences, such as eye, kidney, or skin abnormalities. More than 400 genetic syndromes are thought to be associated with hearing loss. Non-syndromic means that the genetic cause is only associated with hearing loss. It is important to note that some syndromic hearing loss may initially present with only hearing loss (so can mimic non-syndromic) but individuals develop other health concerns later in childhood or adulthood. This is one of the reasons a genetic evaluation with genetic testing is recommended for all children who have congenital hearing loss.
About 70-80% of congenital hearing loss is non-syndromic sensorineural hearing loss (SNHL), while the remaining 20-30% is syndromic. Genetic hearing loss may be inherited in a multiple ways. For non-syndromic hearing loss, it may be inherited in an autosomal recessive (75-80%), autosomal dominant (15-20%), X-linked (<2%), or mitochondrial pattern (<1%). The most common non-syndromic genetic cause is due to pathogenic variants in the GJB2 gene (also called Connexin 26). Other genes that can cause congenital hearing loss include GJB3 (Connexin 31), GJB6 (Connexin 30), and KCNQ4.
What type of genetic or other testing may be recommended?
For infants and children identified to have hearing loss it is important that they undergo a thorough evaluation. In addition to a complete hearing evaluation, a detailed review of their medical history (including prenatal), family history, and physical examination should also be done to help identify the underlying cause for their hearing loss which may aid in establishing appropriate management or treatment approaches.
Multiple organizations have published guidelines for the evaluation of infants and children found to have hearing loss — including the American College of Medical Genetics (ACMG), Joint Committee on Infant Hearing (JCIH), and International Pediatric Otolaryngology Group (IPOG). In summary, it is recommended that:
- CMV testing to be done on all with non-syndromic or sensorineural hearing loss.
- Temporal bone imaging should be considered.
- Genetics evaluation should be offered to families of infants and children with hearing loss in which a environmental cause is not known.
- Genetic testing (multigene panel or single gene testing) should be offered to all patients with sensorineural hearing loss.
- If a genetic syndrome is suspected, genetic testing should be targeted towards the suspected syndrome to confirm diagnosis.
Developmental milestones refer to skills or tasks that most children are able to accomplish by a specific age. Developmental delay is when a child does not meet developmental milestones in one or more areas. This generally refers to situations where someone is significantly and continually delayed, and not to those who are temporarily behind. A child is diagnosed with developmental delay after an assessment that includes evaluation of physical (fine motor skills, gross motor skills), cognitive (intellectual abilities), communication (speech and language), social or emotional development (social skills, emotional control), and adaptive (self-care) skills. The term developmental delay is used in early childhood (before school age or younger than age 5).
Some children will have delays in multiple areas. Children with significant delays in two or more areas are diagnosed with global developmental delay (GDD). Those with GDD are more likely to be diagnosed with intellectual disability (ID) later in childhood. However, not all children with GDD will have ID.
What happens once my child has been diagnosed with developmental delay?
If there are concerns that your child has developmental delays, their primary care physician or pediatrician may recommend further evaluation. This should involve a comprehensive review of your child’s medical history, family history, physical and neurological exam, and may include a referral to meet with a genetics specialist.
Causes of Developmental Delay
Developmental delays may be caused by multiple factors, but for many the cause is unknown. Factors that can contribute include both genetic and environmental (nongenetic) factors (called multifactorial). Nongenetic factors can be things like exposures during the pregnancy, pregnancy or birth complications, poor nutrition, or infections. The chance that there is an underlying genetic condition is increased in those who have GDD and/or have other features or health problems, such as certain physical features, birth defects, hearing loss, or seizures.
What type of genetic testing may be recommended?
For children who have developmental delays, genetic testing may not always be recommended. The decision to proceed with testing may be based on the degree of delays that someone has, and whether they have other features or health problems. For some children, the decision may be made to see how they grow and develop over time, and if there are continued concerns in the future genetic testing may be done.
There are multiple types of tests that may be recommended for those who have developmental delays. The most common include chromosome microarray and fragile X testing. The chromosome microarray is used to identify small missing or extra pieces of chromosome material, and can identify or rule out several hundred known genetic syndromes. Fragile X is a specific condition that can cause developmental delays along with other health issues. Other genetic testing may include single gene testing, multigene panels, or whole exome sequencing, but how useful additional testing like this would be depends on the individual and their specific health concerns.
Intellectual disability (ID) is a developmental disorder where an individual has significant difficulties with intellect and independent functioning that is identified before they turn 18 years old. In the past, intellectual disability has also been called mental retardation and cognitive disability. ID is a spectrum in which individuals may range from being more mildly to more profoundly affected in their abilities. The diagnosis of ID is typically made following the use of a standardized measurements of skills. About 1-3% of the general population is affected by ID.
Causes of Intellectual Disability
Multiple factors can contribute to someone developing ID, and can include both genetic and environmental (nongenetic) influences (called multifactorial). About 40% of those with ID, and up to about 50% of those with moderate to severe ID, will have an underlying genetic cause. Some nongenetic causes of ID include prenatal exposures (e.g. alcohol), infection during pregnancy or in childhood, birth complications, and brain injury or trauma.
For those who have an underlying genetic condition as the cause of their ID, they may also have a chromosome abnormality or single gene disorder (such as PTEN Hamartoma Tumor syndrome) which can involve other health issues. The likelihood of an underlying genetic condition may be impacted by whether a child has other features or health problems, such as distinctive physical features, birth defects, seizures, etc. This is often referred to as “syndromic” ID, and is more likely to have a genetic cause.
What type of genetic testing may be recommended?
There are multiple types of tests that may be recommended for those who have ID. The most common is the chromosome microarray. The chromosome microarray is used to identify small missing or extra pieces of chromosome material, and can identify or rule out several hundred known genetic syndromes. This testing has a detection rate (the chance for the testing to come back positive) of about 10-30% in those with ID.
Further genetic testing that may be considered includes single gene testing, multigene panels, or whole exome sequencing to identify a single gene disorder. Examples of single gene testing include fragile X (accounts for less than 5% of those with ID) or MECP2 gene testing (accounts for less than 2-3% of females with GDD/ID). Through the use of multigene panels and whole exome sequencing, an underlying genetic cause is found in about 30-40% of those with ID.
Other testing that may be recommended includes biochemical screening, which involves measuring the levels of different substances in the blood or urine. These tests are used to look for metabolic conditions that can also cause ID. These type of conditions account for less than 5% of those with ID.
A minor (or soft) marker is a difference that is seen on ultrasound that does not cause any health concerns itself, but increases the chance for certain underlying health conditions. Most of the time, a minor marker is a slight variation in development that is not linked to an underlying health issue.
For example, shorter than average femur (thigh) bones are considered a minor marker that increases the chance for Down syndrome. This is because many babies with Down syndrome have shorter than average thigh bones. But there are also many babies who do NOT have Down syndrome but DO have shorter than average thigh bones. There are also some babies with Down syndrome that have average thigh bones. If a minor marker is found on your ultrasound, it is important to talk with your doctor and/or genetic counselor to understand what that marker means for your pregnancy.
What are some other minor markers that may be seen in the 2nd trimester?
- Shortened or Absent Nasal Bone
- Choroid Plexus Cyst
- Echogenic Intracardiac Focus
- Increased thickness of the nuchal fold
- Renal Pyelectasis/Hydronephrosis (enlarged kidneys)
- Short femur bones
- Short humerus bones
- Single umbilical artery (SUA), also called a two vessel cord
What if a minor marker or multiple markers are seen on my ultrasound?
You should be talk with a medical professional that can provide you more information about what these findings may mean for your baby. Depending on what marker(s) is seen, your doctor may offer you a screening test, such as cell-free DNA, or a diagnostic testing, such as amniocentesis, to look for certain genetic and chromosomal conditions like Down syndrome. It is always a personal choice whether or not to undergo any prenatal testing.
Depending on the markers that are seen on the ultrasound, it may be recommended that you have a follow-up specialized ultrasound to more carefully evaluate the baby’s heart, called a fetal echocardiogram. Babies with chromosome conditions including Down syndrome have a higher chance for a heart defect. If an ultrasound increases the chance for the pregnancy to have a chromosome condition, it may be important in planning your delivery to determine if the baby is likely to have a heart defect.
Receiving a Prenatal Diagnosis
People have very different reactions to receiving a prenatal diagnosis. For some, it represents a chance to know that they will have a child with some special needs ahead of time, allowing them to prepare. For others, they may not feel they are able to care for a child with special needs, and would choose to look into adoption after the baby is born. Others may choose to terminate/abort a pregnancy. This is a very personal and sometimes difficult decision, and is often unexpected. It is important to have access to all of the appropriate information, as well as have the option to discuss these options further with a healthcare provider. Learn more about the various options.
Receiving a prenatal diagnosis is often an unexpected thing to hear during a pregnancy.
GSF recognizes that the decision of what to do next is difficult for many people, and our goal is to make sure you have all of the information you need to make the right decision for you and your family.
If you have decided that you want to continue your pregnancy and raise your baby, know that you are fully supported by professionals in the genetics field in that decision. As you learn more about your baby’s condition and get connected with other families, you will likely find that there is a vast network of support of people who have traveled down this same path and want to be there for you.
Some people may be in a situation where they don’t feel supported in their decision to continue the pregnancy (maybe a partner, extended family members, or a provider has a differing opinion). This can add complexity and strain to a situation that may already be emotionally difficult. It is important to remember that this is your decision, and it is important that you feel as comfortable as possible with that decision.
Finding people to support you on this journey will continue to be very important.
Click below to find more resources for families with a child who has a chromosome or genetic condition:
Receiving a prenatal diagnosis is often an unexpected thing to hear during a pregnancy. GSF recognizes that the decision of what to do next is difficult for many people, and our goal is to make sure you have all of the information you need to make the right decision for you and your family.
If you find out during the pregnancy that the baby has a medical condition, there are several options, including adoption. Raising a child with special needs can be challenging and rewarding (as can raising any child), but it is not the right decision for everyone. If you’ve found out that you have a pregnancy that will have special needs and feel like adoption is something you may consider.
Click below to get more information regarding adoption:
Receiving a prenatal diagnosis is often an unexpected thing to hear during a pregnancy.
GSF recognizes that the decision of what to do next is difficult for many people, and our goal is to make sure you have all of the information you need to make the right decision for you and your family.
Before making a decision to pursue an abortion, we do encourage individuals and couples to make sure that they have all of the information they need to make an informed decision. This is obviously a decision that cannot be undone, and it is important that you are confident in that decision.
If you are at the point where you have made the heart-wrenching and very difficult decision to terminate, you are not alone. We understand that this is a hard decision that every family must make for themselves, and make these decisions for a variety of reasons, and we support you.
One thing that the GSF team has learned over the years is that there are many different religious, cultural, and personal factors that play into this decision. Often times this is a choice that is made in cases where the pregnancy was planned and wanted, which can make this decision even more agonizing.
Your providers should be able to walk you through all of the logistics of this process and support you.
Click below to find additional online resources and support groups: